Biologia plantarum 70:1-10, 2026 | DOI: 10.32615/bp.2026.001
Optimized protocol for in vitro indirect organogenesis and shoot regeneration of Platycladus orientalis
- Faculty of Experimental Sciences, Department of Animal Biology, Plant Biology, and Ecology, University of Jaen, Campus Las Lagunillas s/n, 23071 Jaen, Spain
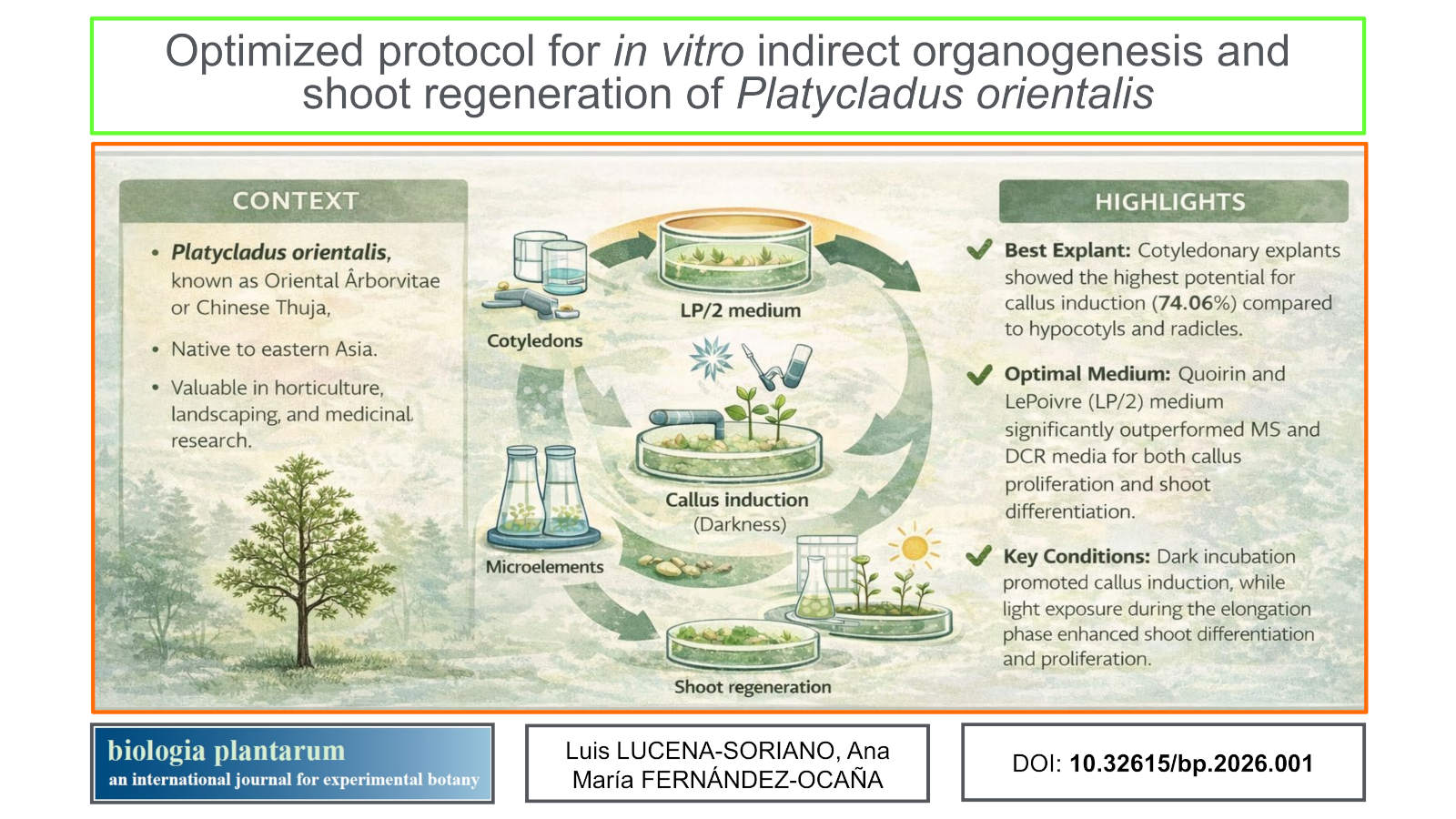
Background: Platycladus orientalis L. is a drought-tolerant conifer valued for its ornamental and medicinal properties. However, efficient regeneration systems for this species remain limited, hindering its propagation and conservation.
Aims: The aim of this study was to develop a reliable protocol for indirect organogenesis of Platycladus orientalis under in vitro conditions, evaluating the influence of explant type, culture medium, light exposure, and pretreatment on regeneration efficiency.
Methods: Cotyledon, hypocotyl, and radicle explants were cultured on different media formulations. The effects of light and darkness during callus induction and shoot elongation were compared. Seeds underwent or avoided vernalization and scarification treatments to assess their influence on germination and callus formation.
Results: Cotyledon explants achieved the highest callus induction rate, reaching 74.06%, particularly under dark conditions. Exposure to light during elongation significantly enhanced callus proliferation and shoots differentiation. Quoirin and LePoivre medium promoted the greatest number of adventitious shoots, with an average of 7.9 shoots per explant, while other media tested showed lower effectiveness. Germination was higher in non-vernalized and non-scarified seeds cultured in Quoirin and LePoivre medium.
Conclusions: The established protocol enables efficient indirect organogenesis and shoot regeneration of Platycladus orientalis using cotyledon explants and Quoirin and LePoivre medium. The finding provides a valuable tool for clonal propagation and conservation of this species, supporting both ornamental cultivation and the preservation of its genetic resources.
Keywords: callus induction, culture medium, indirect organogenesis, in vitro regeneration, Platycladus orientalis, vernalization.
Received: May 16, 2025; Revised: November 15, 2025; Accepted: January 26, 2026; Published online: February 20, 2026 Show citation
| ACS | AIP | APA | ASA | Harvard | Chicago | Chicago Notes | IEEE | ISO690 | MLA | NLM | Turabian | Vancouver |
References
- Ahn, C.-H., & Choi, Y.-E. (2017) In vitro clonal propagation and stable cryopreservation system for Platycladus orientalis via somatic embryogenesis. Plant Cell, Tissue and Organ Culture, 131, 513-523.
 Go to original source...
Go to original source... - Aitken-Christie, J., Singh, A.P. & Davies, H. (1988) Multiplication of meristematic tissue: a new tissue culture system for radiata pine. In: Hanover, J.W., Keathley, D.E., Wilson, C.M. & Kuny, G. (Eds.) Genetic Manipulation of Woody Plants. Basic Life Sciences. Vol. 44. Boston: Springer, pp. 413-432.
 Go to original source...
Go to original source... - Akter, A., Nishida, N., Takada, S. et al. (2018) Genetic and epigenetic regulation of vernalization in Brassicaceae. In: El-Esawi, M.A. (Ed.) Brassica Germplasm - Characterization, Breeding and Utilization. London: IntechOpen, pp. 75-94.
 Go to original source...
Go to original source... - Almqvist, C., Bergsten, U., Bondesson, L., Eriksson, U. (1998) Predicting germination capacity of Pinus sylvestris and Picea abies seeds using temperature data from weather stations. Canadian Journal of Forest Research, 28, 1530-1535.
 Go to original source...
Go to original source... - Ardebol, R.C., de Francisco, L.R., Rosales, Y., Alvarado, O.G. (2006) [In vitro germination of Pinus cubensis Griseb.] Ciencias Holguín, 12, 1-12. [In Spanish]
- Barone, J.O., Luna, C.V. & Oberschelp, G.P.J. (2018) [Effect of basal media on direct organogenesis of Pinus taeda L.] Mendoza: UNCUYO, pp. 7. [In Spanish]
- Barthwal, S., Singh, S., Saha, R., Tadiya, R. & Rawat, S. (2025) Vegetative propagation of forest tree species. In: Mandal, A.K. & Nicodemus, A. (Eds.) Textbook of Forest Science. Singapore: Springer, pp. 679-701.
 Go to original source...
Go to original source... - Bhat, S.J.A., Gangoo, S.A., Geelani, S.M., Qasba, S.S. & Parray A.A. (2014) Callus culture and organogenesis in fir (Abies pindrow Royle). Journal of Cell and Tissue Research, 14, 4653-4658.
- Bonner, F.T. & Brand, G.J. (2004) Platycladus orientalis (L.) Franco - Oriental arborvitae. USDA Forest Service, U.S. Department of Agriculture. Available at: https://www.fs.usda.gov/nsl/Wpsm/Platycladus.pdf.
- Campusano, F.G., González, G.M., Flores, G.G., Mendoza, A.P., Ávila, V.M.C. & Guzmán, J.M. (2019) In vitro adventitious shoot morphogenesis of the Mexican Douglas-fir Pseudotsuga menziesii (Mirb.) Franco. Revista Mexicana de Ciencias Forestales, 10, 22.
- Carneros, E., Celestino, C., Klimaszewska, K., Park, Y.-S., Toribio, M. & Bonga, J.M. (2009) Plant regeneration in stone pine (Pinus pinea L.) by somatic embryogenesis. Plant Cell, Tissue and Organ Culture, 98, 165-178.
 Go to original source...
Go to original source... - Celestino, C., Hernandez, I., Carneros, E., Lopez-Vela, D. & Toribio, M. (2005) Somatic embryogenesis as a main tool in forest biotechnology. Forest Systems, 14, 345-357.
 Go to original source...
Go to original source... - Chang, E., Guo, W., Dong, Y. et al. (2023) Metabolic profiling reveals key metabolites regulating adventitious root formation in ancient Platycladus orientalis cuttings. Frontiers in Plant Science, 14, 1192371.
 Go to original source...
Go to original source... - Cob-Uicab, V.J., Sabja, A.M., Ríos-Leal, D. et al. (2011) Potential of organogenesis as a strategy for the in vitro mass propagation of Fitzroya cupressoides in Austral South America. Revista Chapingo Serie Ciencias Forestales y del Ambiente, 17, 423-433.
 Go to original source...
Go to original source... - Dong, Y., Xiao, W., Guo, W. et al. (2023) Effects of donor ages and propagation methods on seedling growth of Platycladus orientalis (L.) Franco in winter. International Journal of Molecular Sciences, 24, 7170.
 Go to original source...
Go to original source... - Fan, S.-Y., Zeng, H.-W., Pei, Y.-H. et al. (2012) The anti-inflammatory activities of an extract and compounds isolated from Platycladus orientalis (L.) Franco in vitro and ex vivo. Journal of Ethnopharmacology, 141, 647-652.
 Go to original source...
Go to original source... - Furmanek, T. & Banas, W. (2011) Embryogenic callus formation by cotyledon and leaf explants of Crambe abyssinica seedlings. BioTechnologia, 92, 209-213.
 Go to original source...
Go to original source... - Gao, F., Peng, C., Wang, H., Shen, H. & Yang, L. (2021) Selection of culture conditions for callus induction and proliferation by somatic embryogenesis of Pinus koraiensis. Journal of Forestry Research, 32, 483-491.
 Go to original source...
Go to original source... - Gao, F., Shi, Y., Wang, R. et al. (2023) Optimization of key technologies for induction of embryogenic callus and maturation of somatic embryos in Korean pine (Pinus koraiensis). Forests, 14, 850.
 Go to original source...
Go to original source... - Guo, G. & Jeong, B.R. (2021) Explant, medium, and plant growth regulator (PGR) affect induction and proliferation of callus in Abies koreana. Forests, 12, 1388.
 Go to original source...
Go to original source... - Guo, T., Bao, F., Fan, Y., Zhang, J. & Zhao, J. (2024) Small molecules, enormous functions: potential approach for overcoming bottlenecks in embryogenic tissue induction and maintenance in conifers. Horticulture Research, 11, uhae180.
 Go to original source...
Go to original source... - Gupta, P.K. & Durzan, D.J. (1985) Shoot multiplication from mature trees of Douglas-fir (Pseudotsuga menziesii) and sugar pine (Pinus lambertiana). Plant Cell Reports, 4, 177-179.
 Go to original source...
Go to original source... - He, W.T., Hou, S.W. & Wang, C.Y. (2006) Callus induction and high-frequency plant regeneration from hypocotyl and cotyledon explants of Arctium lappa L. In Vitro Cellular & Developmental Biology-Plant, 42, 411-414.
 Go to original source...
Go to original source... - Himanen, K., Nygren, M. & Dumroese, R.K. (2012) Boiling water scarification plus stratification improves germination of Iliamna rivularis seeds. Native Plants Journal, 13, 244-256.
 Go to original source...
Go to original source... - Ho, K.L., Ng, Z.X., Wang, C.W. et al. (2022) Comparative analysis of in vitro enzyme inhibitory activities and phytochemicals from Platycladus orientalis (L.) Franco via solvent partitioning method. Applied Biochemistry and Biotechnology, 194, 3621-3644.
 Go to original source...
Go to original source... - Huapaya, A.E.R. (2015) [Effect of pregermination treatments on irapay seeds (Lepidocaryum tenue Mart.).] Xilema, 28, 79-85. [In Spanish]
- Jacoby, A. (1999) Micropropagation of Pinus species. PhD Thesis. Bloemfontein: University of the Orange Free State, pp. 139.
- Khamushi, M., Dehestani-Ardakani, M., Zarei, A., Aliabad, K.K. (2019) An efficient protocol for micropropagation of old cypress of Abarkuh (Cupressus sempervirens var. horizontalis [Mill.]) under in vitro condition. Plant Cell, Tissue and Organ Culture, 138, 597-601.
 Go to original source...
Go to original source... - Konate, S., Kone, M., Kouakou, H.T., Kouadio, J.Y. & Zouzou, M. (2013) Callus induction and proliferation from cotyledon explants in Bambara groundnut. African Crop Science Journal, 21, 255-263.
- Krasnoperova, V.V. & Bukharina, I.L. (2020) The study into the method of culture in vitro as a method of vegetative propagation of coniferous trees. Russian Agricultural Sciences, 46, 19-22.
 Go to original source...
Go to original source... - Ledea-Rodríguez, J.L., Reyes-Pérez, J.J., Castellanos-Cervantes, T., Angulo, C., Reynoso-Granados, T. & Alcaraz-Meléndez, L. (2020) [In vitro evaluation of germination, development and multiplication of Moringa oleifera Lam in different culture media.] Revista de la Facultad de Agronomía de la Universidad del Zulia, 37, 280-304. [In Spanish]
- Li, G., Du, S. & Wen, Z. (2016) Mapping the climatic suitable habitat of oriental arborvitae (Platycladus orientalis) for introduction and cultivation at a global scale. Scientific Reports, 6, 30009.
 Go to original source...
Go to original source... - Long, Y., Yang, Y., Pan, G., Shen, Y. (2022) New insights into tissue culture plant-regeneration mechanisms. Frontiers in Plant Science, 13, 926752.
 Go to original source...
Go to original source... - Ma, J., Chen, X., Han, F. et al. (2022) The long road to bloom in conifers. Forestry Research, 2, 16.
 Go to original source...
Go to original source... - Maruyama, T.E. & Hosoi, Y. (2019) Progress in somatic embryogenesis of Japanese pines. Frontiers in Plant Science, 10, 31.
 Go to original source...
Go to original source... - Mehbub, H., Akter, A., Akter, M.A. et al. (2022) Tissue culture in ornamentals: cultivation factors, propagation techniques, and its application. Plants, 11, 3208.
 Go to original source...
Go to original source... - Murashige, T. & Skoog, F. (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiologia Plantarum, 15, 473-497.
 Go to original source...
Go to original source... - Orozco-Cardona, A.F., Franco-Herrera, N. & Taborda-Beltrán, L.A. (2010) [Evaluation of three methods of scarification in carob (Hymenaea courbaril L.) seeds.] Revista de Investigaciones de la Universidad del Quindío, 20, 36-41. [In Spanish]
 Go to original source...
Go to original source... - Pais, M.S. (2019) Somatic embryogenesis induction in woody species: the future after OMICs data assessment. Frontiers in Plant Science, 10, 240.
 Go to original source...
Go to original source... - Ramírez Rodas, A.I. (2006) [Adaptation of a somatic embryogenesis method to regenerate asexual embryos of Guatemalan fir (Abies guatemalensis Rehder).] Quetzaltenango: ICTA, pp. 30. [In Spanish]
- Reddy, M.C., Indu, K., Bhargavi, C., Rajendra, M.P., Babu, B.H. (2022) A review on vegetative propagation and applications in forestry. Journal of Plant Development Sciences, 14, 265-272.
- Rix, K.D., Gracie, A.J., Potts, B.M., Brown, P.H., Spurr, C.J., Gore, P.L. (2011) Germination response of Eucalyptus globulus seeds exposed to low and high temperature stress. Seed Science and Technology, 39, 686-691.
 Go to original source...
Go to original source... - Roca, W. & Mroginski, L. (1991) [Tissue Culture in Agriculture: Fundamentals and Applications.] Cali: International Center for Tropical Agriculture, pp. 970. [In Spanish]
- Rojas-Vargas, A., Castander-Olarieta, A., do Nascimento, A.M.M. et al. (2023) Testing explant sources, culture media, and light conditions for the improvement of organogenesis in Pinus ponderosa (P. Lawson and C. Lawson). Plants, 12, 850.
 Go to original source...
Go to original source... - Sarmast, M.K. (2018) In vitro propagation of conifers using mature shoots. Journal of Forestry Research, 29, 565-574.
 Go to original source...
Go to original source... - Shen, X. & Cho, M.-J. (2021) Factors affecting seed germination and establishment of an efficient germination method in sugar pine (Pinus lambertiana Dougl.). HortScience, 56, 299-304.
 Go to original source...
Go to original source... - Shmakov, V.N. & Konstantinov, Yu.M. (2020) Somatic embryogenesis in Larix: the state of the art and perspectives. Vavilov Journal of Genetics and Breeding, 24, 575-588.
 Go to original source...
Go to original source... - Stojičić, D., Budimir, S., Čokeša, V., Uzelac, B. (2024) Optimization of in vitro regeneration of Pinus peuce (Gris.). Horticulturae, 10, 97.
 Go to original source...
Go to original source... - Tang, W. & Newton, R.J. (2005) Plant regeneration from callus cultures derived from mature zygotic embryos in white pine (Pinus strobus L.). Plant Cell Reports, 24, 1-9.
 Go to original source...
Go to original source... - Tang, W., Sederoff, R. & Whetten, R. (2001) Regeneration of transgenic loblolly pine (Pinus taeda L.) from zygotic embryos transformed with Agrobacterium tumefaciens. Planta, 213, 981-989.
 Go to original source...
Go to original source... - Tavares, J.J.D.M. (2019) In vitro morphogenesis assays in Pinus halepensis Mill. MSc Thesis. Coimbra: University of Coimbra.
- Yao, K., Tu, C., Zhang, A., Zeng, Z., Yang, Y. (2025) Distinguishing drought resistance strategies and identifying indicator traits of Platycladus orientalis and Broussonetia papyrifera. Frontiers in Plant Science, 16, 1644756.
 Go to original source...
Go to original source... - Yaroshko, O., Pasternak, T., Larriba, E. & Pérez-Pérez, J.M. (2023) Optimization of callus induction and shoot regeneration from tomato cotyledon explants. Plants, 12, 2942.
 Go to original source...
Go to original source... - Zhu, T., Wang, J., Hu, J., Ling, J. (2022) Mini review: Application of the somatic embryogenesis technique in conifer species. Forestry Research, 2, 18.
 Go to original source...
Go to original source...



