Biologia plantarum 70:38-46, 2026 | DOI: 10.32615/bp.2026.005
Study about factors affecting Agrobacterium-mediated transient expression by vacuum infiltration
- 1 College of Life science, Northwest Normal University, 730070 Lanzhou, People's Republic of China
- 2 Lanzhou Veterinary Research Institute, Chinese Academy of Agricultural Science, 730046 Lanzhou, People's Republic of China
- 3 College of Life science, Lanzhou University, 730070 Lanzhou, People's Republic of China
- 4 New Rural Development Research Institute, Northwest Normal University, 730070 Lanzhou, People's Republic of China
Background: In the last decades, vacuum infiltration had been applied for the Agrobacterium-mediated transient expression of foreign gene in plants. However, the relevant influencing factors have not been fully studied yet.
Aims: This study aimed to evaluate the effects of vacuum infiltration pressure, time of vacuum infiltration, concentration of Agrobacterium tumefaciens (A. tumefaciens), and incubation time post infiltration on the transient expression of foreign gene in leaves of Nicotiana benthamiana (N. benthamiana).
Methods: Two A. tumefaciens strains, LBA4404 and EHA105, carrying a reporter gene of green fluorescent protein (GFP), were used to infiltrate leaves of N. benthamiana via vacuum infiltration. The changes of GFP expression with changes of vacuum infiltration pressure, time of vacuum infiltration, concentration of A. tumefaciens, and incubation time post infiltration were measured.
Results: Increase of vacuum pressure from 0.03 to 0.07 MPa increased the GFP expression in the leaves infiltrated with either LBA4404 or EHA105, and the vacuum pressure at 0.07 MPa made almost all of area of the infiltrated leaf express GFP. The increase of the time of vacuum infiltration from 10 to 20 min significantly enhanced the GFP expression in either the LBA4404 or EHA105-infected leaves. Among the different concentrations of Agrobacterium (from OD600 0.1 to 0.9), the concentrations of LBA4404 strain suspension at OD600 0.5 and EHA105 strain suspension at OD600 0.3, respectively, were most effective concentrations for enhancing the GFP expression in the infected leaves. The increase of incubation time post infiltration from 2 to 4 days largely enhanced the intensity of GFP expression in the either LBA4404 or EHA105-infected leaves. However, further increase of incubation time post infiltration from 4 to 6 days decreased the GFP expression in the infected leaves.
Conclusions: Vacuum infiltration pressure/time, concentration of A. tumefaciens, and the incubation time post infiltration were important factors affecting the level of transient expression by vacuum infiltration. Optimizing these factors is essential for improving the level of transient expression of foreign gene by vacuum infiltration.
Keywords: Agrobacterium tumefaciens, Nicotiana benthamiana, transient expression, vacuum infiltration.
Received: December 11, 2025; Revised: March 12, 2026; Accepted: March 12, 2026; Published online: April 1, 2026 Show citation
| ACS | AIP | APA | ASA | Harvard | Chicago | Chicago Notes | IEEE | ISO690 | MLA | NLM | Turabian | Vancouver |
Introduction
Transgenic plants have emerged as an effective platform for production of recombinant proteins to meet the worldwide demand for the protein-based pharmaceuticals.
The expression of transgene in plants can be achieved by utilizing two different systems: stable genetic transformation or transient gene expression. In stable genetic transformation, the target gene is cloned into the expression vector and delivered to the genomes of nuclear or semiautonomous organelles. The target gene becomes heritable through generations of selected transgenic lines, and transgenic seeds can be stocked for large scale manufacturing of the target protein. However, the process of generation, selection and acquisition of stable transgenic lines is time-consuming (Leuzinger et al., 2013; Chen et al., 2016).
In transient expression in plants, the expression vector harbouring the gene of interest is delivered via gene gun or is delivered via Agrobacterium tumefaciens (A. tumefaciens) to plants through a process called agroinfiltration (Zheng et al., 2012; Krenek et al., 2015). Instead of integrating into the plant genomes, the delivered gene remains episomal and can quickly express the desired protein (Chen et al., 2016). Transient expression in plants can produce the target proteins usually within one week after agroinfiltration, since the delivered gene would be silenced or its expression would be terminated after one week. When using viral vectors, it would take longer time to produce the target proteins because they have to spread throughout the host plant (Lai and Chen, 2012; Chen et al., 2016). Regardless of the methods or expression vectors used for the transient expression in plants, transient expression significantly shortens the time required for the production of the target proteins, compared to stable genetic transformation. Because transient expression in plants is not dependent on stable transgenic lines, transient expression allows easy optimization for the target proteins production. Thus, transient gene expression is more effective and compatible for commercial production of recombinant proteins in plants, compared to stable genetic transformation.
In transient expression in plants at laboratory scale, leaves are infiltrated with the suspension of A. tumefaciens by needle-less syringes (Leuzinger et al., 2013; Chen et al., 2016). However, this method is low efficient and cannot meet the need for large-scale production of recombinant proteins. In addition, using needle-less syringes artificially would cause uneven infiltration, which would affect the subsequent quantification. In recent years, vacuum infiltration has been developed as a more efficient transgene delivery method for transient expression in plants, which can be typically used for larger-scale production of recombinant proteins (Spiegel et al., 2022). In vacuum infiltration of A. tumefaciens, plant leaves are firstly submerged into infiltration media which contains the solution of A. tumefaciens strain harbouring the target recombinant DNA. Vacuum chamber subjects leaves to negative atmospheric pressure that may cause air to be evacuated from the leaf interior through stomata. As vacuum is broken and pressure rapidly returns to initial level, A. tumefaciens may be driven into the leaf interior to replace air (Simmons et al., 2009; Lai and Chen, 2012; Rivera et al., 2012; Chen et al., 2013; Leuzinger et al., 2013; Chen and Lai, 2015; Ashrafi-Dehkordi et al., 2021). This process may easily expose A. tumefaciens to leaf mesophyll cells, making leaves more susceptible to transformation compared with the condition that A. tumefaciens is present in surface of leaves (Tague and Mantis, 2006; Simmons et al., 2009; Diamos et al., 2016; Dattgonde et al., 2019). Thus, researchers believed that vacuum infiltration can largely enhance the efficiency of transient transformation in plants at larger scale for the production of pharmaceutical protein (such as antigen and monoclonal antibody) and biosynthesis of plant natural product (such as terpenoid) by expressing the genes encoding the pharmaceutical proteins or enzymes of biosynthetic pathway (Chen et al., 2013; Reed and Osbourn, 2018). For example, vacuum infiltration has been used for transient expression of the recombinant proteins in some plant species, such as in Nicotiana benthamiana (N. benthamiana) for expressing blood-stage malaria antigens (Boes et al., 2016) and in Mucuna bracteata for expressing anti-toxoplasma immunoglobulin ( Abd-Aziz et al., 2020). The Medicago Inc. (Canada) has developed the COVID-19 vaccine (authorized by Health Canada in 2022) and virus-like particles vaccine for influenza (submitted to the FDA for approval in 2025) in N. benthamiana by using vacuum infiltration (https://www.medicago.com/en/discovery). Thus, transient expression by vacuum infiltration of plants is a promising production system for fighting the diseases of human and animals.
As introduced above, the Agrobacterium-mediated transient expression by vacuum infiltration is a complex process, including A. tumefaciens infection through vacuum infiltration, and protein biosynthesis and accumulation in the leaf tissue. Whether or to what extent the factors or parameters involved in the Agrobacterium-mediated transient expression by vacuum infiltration, including type and concentration of A. tumefaciens, pressure of vacuum infiltration, and incubation time post infiltration, would affect the efficiency of transient gene expression is largely unknown. However, this information could be particularly important for optimizing transient expression via vacuum infiltration in plants to achieve the highest efficiency.
In the present work, under the condition of N. benthamiana leaves infected by two different A. tumefaciens strains (LBA4404 and EHA105), we evaluated the effects of vacuum infiltration pressure, vacuum infiltration time, A. tumefaciens concentration, and incubation time post infiltration on the transient expression in the leaves of N. benthamiana via green fluorescent protein (GFP) as a reporter. We believe that this study would provide valuable information for the studies about the transient expression by vacuum infiltration in plants.
Material and methods
Plant material: Two N. benthamiana seeds were planted into each peat pellet, the tray was covered with a transparent plastic dome, and placed at 25°C with 50% humidity. The dome was removed two weeks later and plants were grown in a greenhouse at 25℃ with a 16 h light/8 h dark cycle. After the water in the tray was drained, plants were irrigated with a Jack's fertilizer at a concentration of 7.4 g L-1. The 3-week-old plants were transferred to a new tray to provide adequate space for further growth until they are ready to be infiltrated in the 4th week of growth.
The geminivirus-derived expression vector: The expression vector based on bean yellow dwarf virus (BeYDV) was presented by Professor Hugh S. Mason of Arizona State University. An enhanced GFP (S65T GFP) gene derived from Aequorea victoria was constructed downstream of the 35S promoter and upstream double terminators (EU and NbACT3) in the vector as the reporter gene. The expression vector based on BeYDV was transformed into Agrobacterium LBA4404 and EHA105 strain via heat shock, and the antibiotic resistance gene of the vector was used for screening and subculture.
Preparation of Agrobacterium cultures: Agrobacterium LBA4404 strain harboring the expression vector with the GFP gene was streaked on YEB agar plates containing kanamycin (50 μg mL-1), rifampicin (25 μg mL-1) and chloramphenicol (25 μg mL-1). Similarly, the EHA105 strain harbouring the same vector was streaked on YEB agar plates containing kanamycin (50 μg mL-1) and rifampicin (25 μg mL-1). After 24 h growth, the single colonies were picked out from the YEB agar plate and inoculated into 400 mL YEB liquid medium with antibiotics, at 28℃ for 48 h in a shaker at 180 rpm. The bacteria were collected by centrifugation at 2 392 × g for 10 min and then washed by infiltration buffer (10 mM MES-KOH, pH 5.5; 10 mM MgSO4; 100 μM acetosyringone). The bacteria were then collected again and resuspended with infiltration buffer. The bacterial concentrations were determined by measuring optical density (OD) at 600 nm and were diluted to the concentrations needed.
Vacuum infiltration: A bass and oil-free vacuum pump (VP750-30L, DAERTUO, Jiangsu Province, China) was used in this work, to which a desiccator was attached (Fig. 1A). The vacuum desiccator was washed with distilled water, then 1 L of Agrobacterium suspension was transferred into the vacuum desiccator. Three plants were transferred into the vacuum desiccator, in which the entire leaf was submerged into the infiltration buffer (Fig. 1B-D). The desiccator was connected to vacuum pump with vacuum tube. After the vacuum pump was turned on, the intake valve was opened and the suction valve was closed. The vacuum negative pressure reached the set value needed and was maintained for certain time (see the part of “experimental sets”). After then, the release valve was slowly opened during certain time (see the part of “experimental sets”) to allow bacterial entry into the interstitial spaces of submerged plant tissue. After agroinfiltration, the plants were transferred back to the growth room for GFP expression and accumulation.

Fig. 1. Vacuum infiltration of N. benthamiana leaves with Agrobacterium suspension. (A) Agrobacterium LBA4404 and EHA105 strains harboring the expression vector with the GFP gene were resuspended with 1 L infiltration buffer and loaded into desiccator; (B) The desiccator was connected to a vacuum pump, and agroinfiltration was operated by applying and releasing vacuum; (C,D) Three plants were placed upside down on the supporting ring, and the entire leaf was submerged into the infiltration buffer.
Experimental sets: In the first set of experiment, Agrobacterium concentration of LBA4404 and EHA105 strain was set at OD600 0.5 and 0.3, respectively. Vacuum infiltration pressure was set at 0.03, 0.05, and 0.07 MPa, respectively. The vacuum infiltration time was set at 15 min (vacuum for 10 min and then slowly releasing vacuum for 5 min). After vacuum infiltration, the infected seedlings were incubated for 4 d under the conditions as described above.
In the second set of experiment, Agrobacterium concentration of LBA4404 and EHA105 strain infection, was set at OD600 0.5 and 0.3, respectively. Vacuum infiltration pressure was set at 0.07 MPa. The vacuum infiltration time was set at 10 min (vacuum for 5 min and then slowly releasing vacuum for 5 min), 15 min (vacuum for 10 min and then slowly releasing vacuum for 5 min), and 20 min (vacuum for 15 min and then slowly releasing vacuum for 5 min). After vacuum infiltration, the infected seedlings were incubated for 4 d under the conditions as described above.
In the second set of experiment, Agrobacterium concentration of LBA4404 and EHA105 strain infection, was set at OD600 0.5 and 0.3, respectively. Vacuum infiltration pressure was set at 0.07 MPa. The vacuum infiltration time was set at 10 min (vacuum for 5 min and then slowly releasing vacuum for 5 min), 15 min (vacuum for 10 min and then slowly releasing vacuum for 5 min), and 20 min (vacuum for 15 min and then slowly releasing vacuum for 5 min). After vacuum infiltration, the infected seedlings were incubated for 4 d under the conditions as described above.
In the third set of experiment, Agrobacterium concentration of LBA4404 and EHA105 strain was set at OD600 0.1, 0.3, 0.5, 0.7 and 0.9, respectively. Vacuum infiltration pressure was set as 0.07 MPa. The vacuum infiltration time was set at 15 min (vacuum for 10 min and then slowly releasing vacuum for 5 min). After vacuum infiltration, the infected seedlings were incubated for 4 d under the conditions as described above.
In the fourth set of experiment, Agrobacterium concentration of LBA4404 and EHA105 strain infection, was set at OD600 0.5 and 0.3, respectively. The vakuum infiltration pressure was set as 0.07 MPa. The vacuum infiltration time was set at 15 min (vacuum for 10 min and then slowly releasing vacuum for 5 min). After vacuum infiltration, the infected seedlings were incubated for 2, 4, and 6 d, respectively, under the conditions as described above.
GFP fluorescent protein detection and photography: The agroinfiltrated leaves were examined for green fluorescence to determine whether the GFP reporter gene had been stably expressed. For the photograph of the whole leaf, the agroinfiltrated leaf was moved into Cabin type ultraviolet analyzer and excited by UV illumination (365 nm) on the back side of infiltrated leaf. The whole leaf was photographed with a regular digital camera. For the photograph of the leaf zone-having GFP expression, the lower surface of the infiltrated leaf was placed under a Leica fluorescent stereomicroscope (Leica Microsystems Ltd., DFC450 C). The infiltrated leaf was excited at a wavelength of 450 - 490 nm and the emission spectrum between 500 - 550 nm was recorded (Stahl et al., 2013).
Quantitative detection of GFP fluorescence was performed according to Chen et al., 2003. The leaf samples producing GFP were fully homogenized with 1:5 (w/v) pre-cooled extraction buffer (30 mM Tris-HCl, 10 mM EDTA, 10 mM NaCl, 5 mM dithiothreitol pH = 8). The homogenate was centrifuged at 15 000 × g for 15 min (4℃). The intensity of green fluorescence emission was measured with the excitation wavelength of 485 nm and the emission wavelength of 538 nm, respectively. Agrobacterium without GFP gene was used to infiltrate and infect leaves, and the intensity of fluorescence emission was measured by the same method, which was taken as a background value and was subtracted from the sample.
Statistical analysis: The data were subjected to analysis of variance (ANOVA) with IBM SPSS Statistics 26 software (SPSS Inc., Chicago, IL, USA), comparisons between the mean values of treatments were done by the Duncan's multiple range test at the confidence level of P < 0.05. The bar chart is made with Origin Pro 9.1 software (OriginLab Corp., Northampton, MA, USA) according to the experimental data analysis, and variability of the data was expressed as mean ± standard deviation (SD) from at least 5 biological repeats.
Results
Effects of pressure of vacuum infiltration on transient expression: As described in the first set of experiment (see the part of “experimental sets”), we firstly studied the effects of pressure of vacuum infiltration on the efficiency of transient expression. The result showed that vacuum infiltration pressure at 0.03 MPa only made partial zone of the infiltrated leaf present green fluorescence from GFP. Increase of vacuum infiltration pressure from 0.03 to 0.05 MPa obviously increased the area having green fluorescence in the total area of the infiltrated leaf. When vacuum pressure reached 0.07 MPa, almost all area of the infiltrated leaf presented green fluorescence (Fig. 2A).
In the infiltrated leaves exposed to different vacuum infiltration pressure, we also measured the expression of GFP in leaves. In the leaves infected by two types of Agrobacterium (LBA4404 and EHA105), the expression of GFP increased with the increase of vacuum pressure from 0.03 to 0.05 MPa. When the vacuum pressure value increased from 0.05 to 0.07 MPa, the expression of GFP in the leaves increased further (Fig. 2B-D).
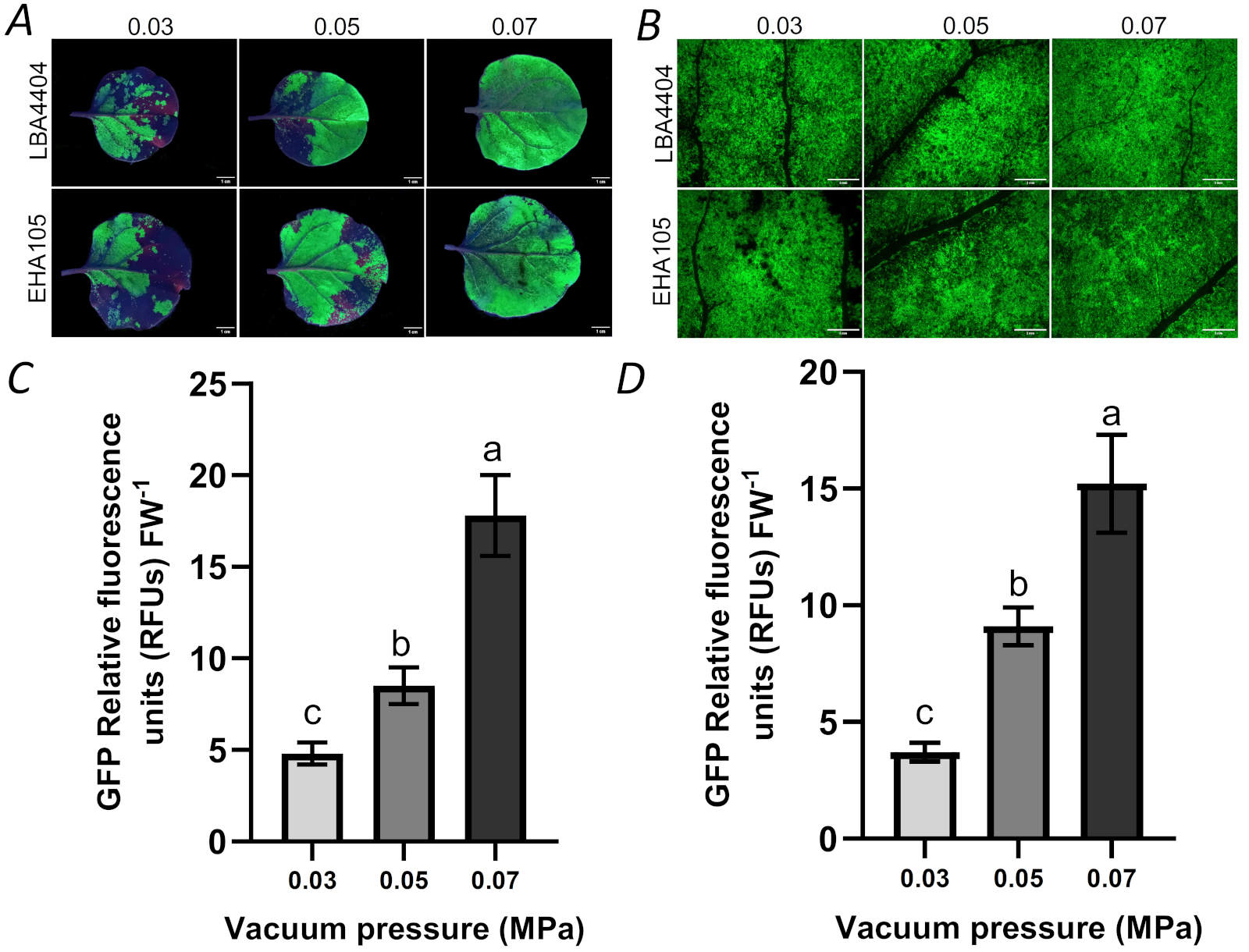
Fig. 2. The effects of vacuum pressure (0.03, 0.05, 0.07 MPa, respectively) on the expression of GFP by vacuum infiltration. N. benthamiana leaves were infiltrated with Agrobacterium EHA105 and LBA4404 harboring the GFP gene. (A) N. benthamiana leaves were infiltrated with EHA105 and LBA4404 harboring the GFP gene, bar = 1 cm; (B) Zone having GFP expression from the agroinfiltrated leaf presented in Fig. 2A was photographed with fluorescent stereomicroscope, bar = 3 mm; (C) The intensity of relative GFP fluorescence in the LBA4404-infiltrated leaf presented with different vacuum pressure; (D) The intensity of relative GFP fluorescence in the EHA105-infiltrated leaf presented with different vacuum pressure. FW - fresh weight. Means ± SD of 5 individual replications at least. Different letters indicate statistically significant differences according to Duncan's multiple range test (P < 0.05).
Effect of time of vacuum infiltration on transient expression: As described in the second set of experiment (see the part of “experimental sets”), we also investigated the effects of the time of vacuum infiltration on transient expression of GFP gene. For the leaves infiltrated by LBA4404 or EHA105, the expression of GFP in the leaves increased with the time of infiltration, but there was no significant difference between the 10 min and 15 min of the vacuum infiltration. Compared with the vacuum infiltration for 15 min, GFP expression in the leaves increased further after 20 min of vacuum infiltration, but there was still no significant difference. Compared with 10 min of vacuum infiltration, GFP expression in the leaves infected by two types of Agrobacterium (LBA4404 and EHA105) increased after 20 min of vacuum infiltration significantly (Fig. 3).
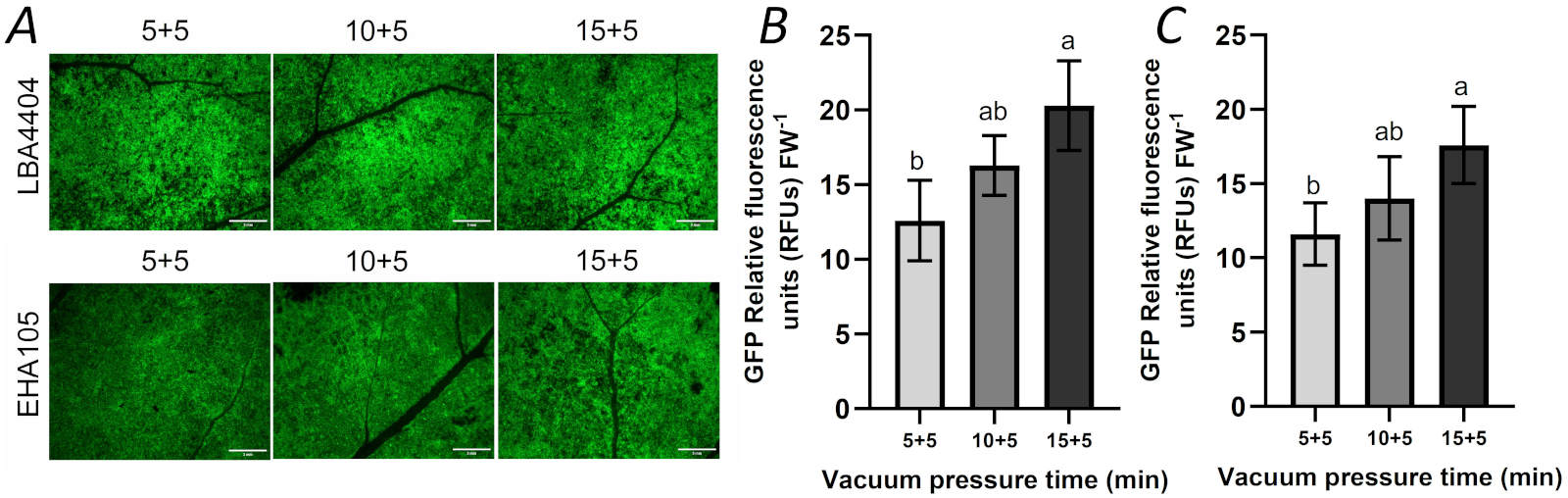
Fig. 3. The effects of the time of vacuum infiltration (10, 15, and 20 min, respectively) on the expression of GFP by vacuum infiltration. (A) Zone having GFP expression from the agroinfiltrated leaf with different vacuum infiltration times was photographed with fluorescent stereomicroscope; (B) Intensity of relative GFP fluorescence in the LBA4404-infiltrated leaf with different vacuum infiltration times; (C) The intensity of relative GFP fluorescence in the EHA105-infiltrated leaf with different vacuum infiltration times. Infiltration times - 10 min (vacuum for 5 min and slowly releasing vacuum for 5 min), 15 min (vacuum for 10 min and slowly releasing vacuum for 5 min), 20 min (vacuum for 15 min and slowly releasing vacuum for 5 min). Bar = 3 mm. FW - fresh weight. Means ± SD of 5 individual replications at least. Different letters indicate statistically significant differences according to Duncan's multiple range test (P < 0.05).
Effects of Agrobacterium concentration on transient expression:
As described in the third set of experiment (see the part of “experimental sets”), we further investigated the effects of different concentrations of the LBA4404 and EHA105 on transient expression. With the increase of the concentration of the LBA4404 from 0.1 to 0.9 (OD600), the GFP expression also presented a pattern that increased first and then decreased. The GFP expression peaked when the concentration of the LBA4404 was at 0.5 (OD600) (Fig. 4A,B). With the increase of the concentration of the EHA105 from 0.1 to 0.9, the GFP expression also presented a pattern that increased first and then decreased. However, different from LBA4404, the GFP expression in EHA105 infected leaves peaked when the concentration of EHA105 was at 0.3 (OD600) (Fig. 4A,C).
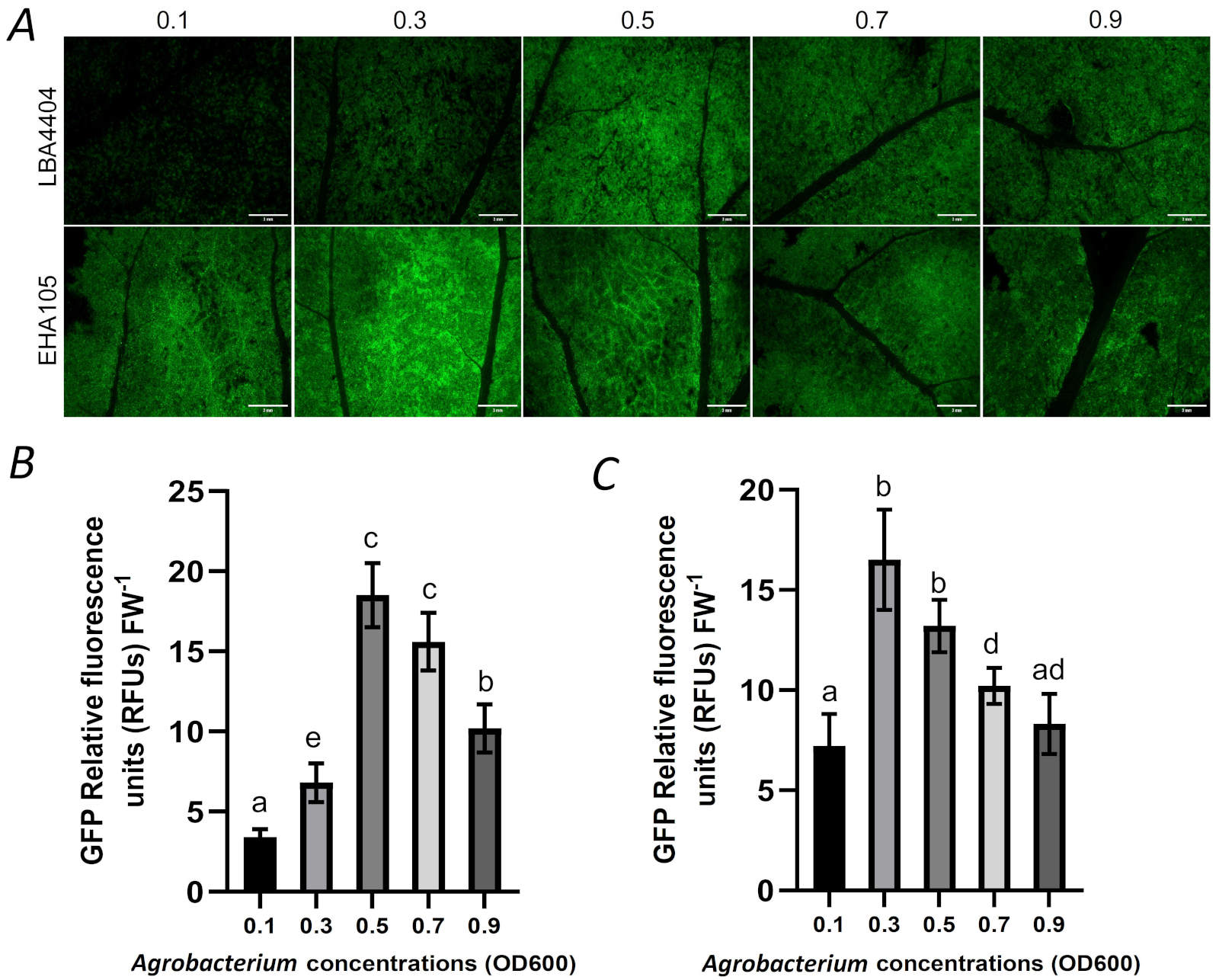
Fig. 4. The effects of Agrobacterium concentrations (OD600 0.1 to 0.9) on the expression of GFP by vacuum infiltration. (A) Zone having GFP expression from the agroinfiltrated leaf with different Agrobacterium concentrations was photographed with fluorescent stereomicroscope; (B) The intensity of relative GFP fluorescence in the LBA4404-infiltrated leaf with different Agrobacterium concentrations; (C) The intensity of relative GFP fluorescence in the EHA105-infiltrated leaf with different Agrobacterium concentrations. Bar = 3 mm. FW - fresh weight. Means ± SD of 5 individual replications at least. Different letters indicate statistically significant differences according to Duncan's multiple range test (P < 0.05).
Effect of incubation time post infiltration on transient expression: The incubation time post infiltration is required for the accumulation of recombinant protein. As described in the fourth set of experiment (see the part of “experimental sets”), we also studied the effects of the incubation days post infiltration (dpi) on transient expression of GFP, which was produced after the infection by LBA4404 and EHA105 strains. The results showed that the increase of incubation time from 2 to 4 d obviously enhanced the level of the GFP expression by LBA4404 infection. However, further increase of incubation time from 4 to 6 days decreased the level of the fluorescent intensity in the GFP expression (Fig. 5A,B). The effect of incubation time post infiltration on GFP expression by EHA105 infection was similar to that of LBA4404 infected leaves (Fig. 5A,C).
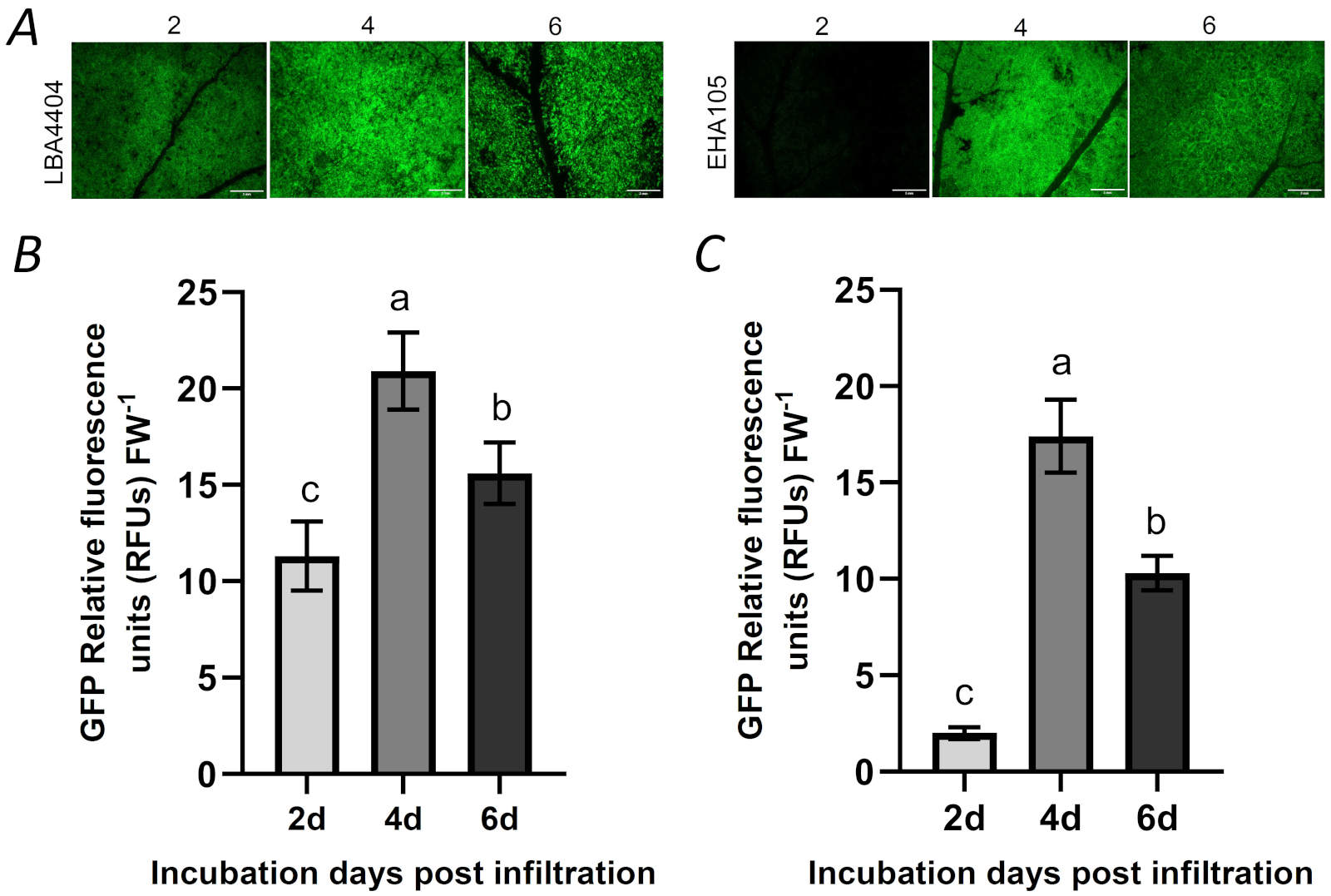
Fig. 5. The effects of incubation time (2, 4, 6 days) post infiltration on the expression of GFP by vacuum infiltration. (A) Zone having GFP expression from the agroinfiltrated leaf with different incubation days post infiltration was photographed with fluorescent stereomicroscope (2d, 4d, 6d: 2, 4, 6 days post infiltration, respectively); (B) The intensity of relative GFP fluorescence in the LBA4404-infiltrated leaf with different incubation days post infiltration; (C) The intensity of relative GFP fluorescence in the LBA4404-infiltrated leaf with different incubation days post infiltration. Bar = 3 mm. FW - fresh weight. Means ± SD of 5 individual replications at least. Different letters indicate statistically significant differences according to Duncan's multiple range test (P < 0.05).
Discussion
Compared with injection, value of vacuum infiltration in the plant transient expression lies in its enormous scalability potential, which is more robust and can infiltrate more plant materials in a short period of time. However, whether some changes of the factors that are involved in the Agrobacterium-mediated transient transformation by vacuum infiltration would affect the transient expression is still largely unknown.
Different works used the seedlings with different growth period for transient expression by vacuum infiltration. For example, Marques et al. (2020) employed N. benthamiana seedlings grown for 6 to 7 weeks, while Stephenson et al. (2018) employed 5-week-old N. benthamiana seedlings for transient expression. Based on our previous work (Zhang et al., 2023), 4~5-week-old N. benthamiana seedlings had the highest level of transient expression of GFP among the seedlings grown for 4 - 8 weeks. Combining this with the perspective of saving the growth time of the seedlings, 4-week-old seedlings could be most appropriate for rapid production of recombinant protein and thus were chosen in the present work to study the effects of vacuum infiltration pressure, time of vacuum infiltration, concentration of A. tumefaciens, and incubation time on transient expression.
In some works using different plant species or with different growth period, different rounds (from 1 to 3 rounds, even more) of vacuum infiltration were performed to enable high efficiency of transformation (Tague and Mantis, 2006; Simmons et al., 2009; Adesoye et al., 2010; King et al., 2015; Koudounas et al., 2022; Trull et al., 2024; Fuhrmann-Aoyagi et al., 2024), possibly considering that the sensitivity to the vacuum-based infiltration with A. tumefaciens would be different among different plant species or would change with changes of anatomy of leaf tissues at different developmental stages. However, multi-rounds of vacuum infiltration are little carried out in N. benthamiana, possibly due to that N. benthamiana is highly susceptible to A. tumefaciens and thus one round of vacuum infiltration seems to be sufficient for high efficiency of transient expression. It has been also reported that young N. benthamiana plants were more susceptible to pathogen than the older ones (Shibata et al., 2010). Based on these reports and the perspective of simplifying the procedure, our present protocol used one round of vacuum infiltration to transiently express GFP in the 4-week-old N. benthamiana seedlings. The observations showed that, with one round of vacuum infiltration, the increase of vacuum pressure from 0.03 to 0.07 MPa increased the area having GFP expression in the total area of the infiltrated leaf, and the vacuum pressure at 0.07 MPa made almost all area of the infiltrated leaf present green fluorescence (Fig. 2). It is possible that increase of vacuum pressure could make more stomata open or make more wounding in the leaf surface, thus increasing the area of leaf surface that can be infected by Agrobacterium strain.
Previous works also showed that the type of bacterial strain is a significant factor for target protein expression (Selvaraj et al., 2010; Liu et al., 2019). Researchers think that, depending on the target plant species and the interaction between Agrobacterium and its host plants, different Agrobacterium strains differ in their virulence. Such variations could be caused by differences in chromosomal background, abilities of the Agrobacterium strains for attachment to plant cells, and the mechanism for the T-DNA delivery (Wroblewski et al., 2005; Baranski et al., 2006). We noted that under the vacuum pressure of 0.03 MPa, the expression of GFP in LBA4404 infected leaves was significantly higher than that in EHA105 infection, which also reflected that the type of Agrobacterium strain was an important factor affecting the expression of foreign genes. However, when the vacuum pressure was 0.05 MPa and 0.07 MPa, there was no significant difference in the expression of GFP in the two kinds of the Agrobacterium infected leaves (Fig. 2C). This observation suggests that the effect of vacuum pressure on foreign genes expression exceeds the effect of Agrobacterium strain type at higher vacuum pressure values.
As introduced above, vacuum can cause the air to be evacuated from the leaf interior. After then, it is needed that vacuum pressure is released so that Agrobacterium can be driven into the leaf interior to replace the air. In theory, increase in the time for the vacuum producing negative pressure could make more air evacuated from the leaf interior and thus affect the subsequent entry of Agrobacterium into plant tissue. However, the results of this study showed that increasing the vacuum pressure for 5 min was not enough to improve the instantaneous expression level of genes, while increasing the vacuum pressure for 10 min could improve the instantaneous expression level of genes (Fig. 3).
Studies have shown that the increase of A. tumefaciens concentration can increase the instantaneous level of genes (Nanasato et al., 2013; Ashrafi-Dehkordi et al., 2021). However, our study found that GFP expression in the leaves infected by LBA4404 and EHA105 showed a trend of first increase and then decrease with the increase of A. tumefaciens concentration (Fig. 4). It can be seen that the appropriate concentration of A. tumefaciens is more conducive to the expression of foreign genes than the high concentration of A. tumefaciens. Studies have shown that A. tumefaciens is also a plant pathogen that has complex effects on infiltrated leaf tissues and often elicits a cell death response (Diamos et al., 2016). In fact, A. tumefaciens can trigger leaf chlorosis or wilting a few days after infiltration (Wroblewski et al., 2005; Nanasato et al., 2013). In addition, A. tumefaciens also survives in leaves for a relatively long time, which could also affect the condition of plants. For example, A. tumefaciens would disrupt metabolism of its host plants via consuming nutrients or other manners (Gonzalez-Mula et al., 2019). Thus, we suggest that, although the moderate concentration of Agrobacterium suspension contains less amount of target genes compared to the higher concentration of Agrobacterium suspension, this can reduce the level of plant cell death in the Agrobacterium-infected tissues. In theory, intensity of GFP expression in the plant tissues is determined not only by the number of gene copies but also by cell viability and plant metabolism (Lai and Chen, 2012). Thus, under the moderate concentration of Agrobacterium suspension, the number of gene copies and cell viability of plant cells could get the best balance point to purse the highest level of intensity of GFP expression.
Our results showed that GFP expression increased markedly when incubation time post infiltration was extended from 2 to 4 days, but then declined when dpi was further increased from 4 to 6 days (Fig. 5). In comparison, the time course of GFP accumulation presented by our work is earlier than those in some other reports. For example, Lombardi et al. (2009) showed that the transient expression of HIV-1 Nef antigens in the 6-week-old N. benthamiana seedlings by the vacuum-based agroinfiltration was detected and increased from 3 to 9 dpi, and the expression level was higher in middle and bottom leaves than that in the youngest top leaves. Using 6-week-old N. benthamiana seedlings and the cowpea mosaic virus (CPMV)-based vector, Loh and Wayah (2014) found that the GFP expression by the vacuum-based agroinfiltration was detectable and increased from 2 to 10 dpi, with the peak at 8 - 10 dpi. This difference of the time course of transient expression of recombinant proteins could depend on the complexity of the expressed proteins, age of the plants, vectors types, and position of the leaf. In our works, the shorter growth period of the N. benthamiana seedlings (the 4-week-old) could be main reason for the earlier peak of GFP expression level than those in some other reports. In the production of biopharmaceuticals, larger and older plants were used for the need for biomass yield and protein accumulation. But, the relatively young plants seem to quicken the time for the accumulation of recombinant proteins and plant growth. Thus, relatively young plants can also be considered for the optimal plant material that balances the need for biomass yield, protein accumulation, space requirements, and time saving.
It has been reported that during transgene expression in plants, the foreign gene may be lost through cell division with the growth of plants, which could decrease the efficiency of transient expression in plants (Kroll and Rathert, 2018). Therefore, the decrease of GFP expression when dpi increased from 4 days to 6 days may be related to the progressive loss of GFP coding gene. Otherwise, post-transcriptional gene silencing (PTGS), which is known as a natural defense mechanism relying on RNA interference for plants against pathogens, often occurs in plants to reduce the expression level of foreign protein through degradation and silencing of the transgene (Wassenegger and Pélissier, 1998; Meins, 2000; Bazzini et al., 2007). Thus, the decrease of GFP expression at 6 dpi may also be related to the activation of PTGS. Since the BeYDV-based expression vector, which is used by the present work, have been reported to cause leaf necrosis, it is also reasonable to assume that the further increase of incubation time post infiltration could cause more cell death of plant tissues, thus decreasing the level of intensity of GFP expression in the plant tissues. It is exciting that modest reduction in expression of Rep and RepA of the BeYDV-based expression vector can reduce plant leaf cell death and increases target protein accumulation (Diamos and Mason, 2019).
In conclusion, the types of Agrobacterium strains, vacuum infiltration pressure, vacuum infiltration time, the concentration of bacteria, and incubation time post infiltration all affected the expression level of transient foreign genes in plants to varying degrees. Optimizing the above factors is of great significance for the improvement of transient expression level of foreign genes.
Download citation
References
- Abd-Aziz, N., Tan, B.C., Rejab, N.A., Othman, R.Y. & Khalid, N. (2020) A new plant expression system for producing pharmaceutical proteins. Molecular Biotechnology, 62, 240-251.
 Go to original source...
Go to original source... - Adesoye, A.I., Togun, A.O., Machuka, J. (2010) Transformation of cowpea (Vigna unguiculata L. Walp.) by Agrobacterium infiltration. Journal of Applied Biosciences, 30, 1845-1860.
- Ashrafi-Dehkordi, E., Alemzadeh, A., Tanaka, N. & Razi, H. (2021) Effects of vacuum infiltration, Agrobacterium cell density and acetosyringone concentration on Agrobacterium-mediated transformation of bread wheat. Journal of Consumer Protection and Food Safety, 16, 59-69.
 Go to original source...
Go to original source... - Baranski, R., Klocke, E. & Schumann, G. (2006) Green fluorescent protein as an efficient selection marker for Agrobacterium rhizogenes mediated carrot transformation. Plant Cell Reports, 25, 190-197.
 Go to original source...
Go to original source... - Bazzini, A.A., Mongelli, V.C., Hopp, H.E., del Vas, M. & Asurmendi, S. (2007) A practical approach to the understanding and teaching of RNA silencing in plants. Electronic Journal of Biotechnology, 10, 178-190.
 Go to original source...
Go to original source... - Boes, A., Reimann, A., Twyman, R.M., Fischer, R., Schillberg, S. & Spiegel, H. (2016) A plant-based transient expression system for the rapid production of malaria vaccine candidates. In: Thomas, S. (Ed.) Vaccine Design. Methods in Molecular Biology. Vol. 1404. New York: Humana, pp. 597-619.
 Go to original source...
Go to original source... - Chen, N., Hsiang, T. & Goodwin, P.H. (2003) Use of green fluorescent protein to quantify the growth of Colletotrichum during infection of tobacco. Journal of Microbiological Methods, 53, 113-122.
 Go to original source...
Go to original source... - Chen, Q. & Lai, H. (2015) Gene delivery into plant cells for recombinant protein production. BioMed Research International, 2015, 932161.
 Go to original source...
Go to original source... - Chen, Q., Dent, M., Hurtado, J. et al. (2016) Transient protein expression by agroinfiltration in lettuce. In: MacDonald, J., Kolotilin, I. & Menassa, R. (Eds.) Recombinant Proteins from Plants. Methods in Molecular Biology. Vol. 1385. New York: Humana Press, pp. 55-67.
 Go to original source...
Go to original source... - Chen, Q., Lai, H., Hurtado, J., Stahnke, J., Leuzinger, K. & Dent, M. (2013) Agroinfiltration as an effective and scalable strategy of gene delivery for production of pharmaceutical proteins. Advanced Techniques in Biology & Medicine, 1, 1000103.
 Go to original source...
Go to original source... - Dattgonde, N., Tiwari, S., Sapre, S. & Gontia-Mishra, I. (2019) Genetic transformation of oat mediated by Agrobacterium is enhanced with sonication and vacuum infiltration. Iranian Journal of Biotechnology, 17, 68-73.
 Go to original source...
Go to original source... - Diamos, A.G. & Mason, H.S. (2019) Modifying the replication of geminiviral vectors reduces cell death and enhances expression of biopharmaceutical proteins in Nicotiana benthamiana leaves. Frontiers in Plant Science, 9, 1974.
 Go to original source...
Go to original source... - Diamos, A.G., Rosenthal, S.H. & Mason, H.S. (2016) 5' and 3' untranslated regions strongly enhance performance of geminiviral replicons in Nicotiana benthamiana leaves. Frontiers in Plant Science, 7, 200.
 Go to original source...
Go to original source... - Fuhrmann-Aoyagi, M.B., Igarashi, S. & Miura, K. (2024) Comparative evaluation of transient protein expression efficiency in tissues across soybean varieties using the Tsukuba system. Plants, 13, 858.
 Go to original source...
Go to original source... - Gonzalez-Mula, A., Lachat, J., Mathias, L. et al. (2019) The biotroph Agrobacterium tumefaciens thrives in tumors by exploiting a wide spectrum of plant host metabolites. New Phytologist, 222, 455-467.
 Go to original source...
Go to original source... - King, J.L., Finer, J.J. & McHale, L.K. (2015) Development and optimization of agroinfiltration for soybean. Plant Cell Reports, 34, 133-140.
 Go to original source...
Go to original source... - Koudounas, K., Carqueijeiro, I., Lemos Cruz, P. et al. (2022) A rapid and efficient vacuum-based agroinfiltration protocol for transient gene overexpression in leaves of Catharanthus roseus. In: Courdavault, V., Besseau, S. (Eds.) Catharanthus roseus. Methods in Molecular Biology. Vol. 2505. New York: Humana, pp. 263-279.
 Go to original source...
Go to original source... - Krenek, P., Samajova, O., Luptovciak, I., Doskocilova, A., Komis, G. & Samaj, J. (2015) Transient plant transformation mediated by Agrobacterium tumefaciens: principles, methods and applications. Biotechnology Advances, 33, 1024-1042.
 Go to original source...
Go to original source... - Kroll, C. & Rathert, P. (2018) Stable expression of epigenome editors via viral delivery and genomic integration. In: Jeltsch, A., Rots, M. (Eds.) Epigenome Editing. Methods in Molecular Biology. Vol. 1767. New York: Humana Press, pp. 215-225.
 Go to original source...
Go to original source... - Lai, H. & Chen, Q. (2012) Bioprocessing of plant-derived virus-like particles of Norwalk virus capsid protein under current Good Manufacture Practice regulations. Plant Cell Reports, 31, 573-584.
 Go to original source...
Go to original source... - Leuzinger, K., Dent, M., Hurtado, J. et al. (2013) Efficient agroinfiltration of plants for high-level transient expression of recombinant proteins. Journal of Visualized Experiments, 77, e50521.
 Go to original source...
Go to original source... - Liu, K., Yang, Q., Yang, T. et al. (2019) Development of Agrobacterium-mediated transient expression system in Caragana intermedia and characterization of CiDREB1C in stress response. BMC Plant Biology, 19, 237.
 Go to original source...
Go to original source... - Loh, H.-S. and Wayah, S.B. (2014) Optimizations of laboratory-scale vacuum-assisted agroinfiltration for delivery of a transgene in Nicotiana benthamiana. Asian Journal of Biotechnology, 6, 1-14.
 Go to original source...
Go to original source... - Lombardi, R., Circelli, P., Villani, M.E. et al. (2009) High-level HIV-1 Nef transient expression in Nicotiana benthamiana using the P19 gene silencing suppressor protein of Artichoke Mottled Crinckle Virus. BMC Biotechnology, 9, 96.
 Go to original source...
Go to original source... - Marques, L.É., Silva, B.B., Dutra, R.F., Florean, E.O.T., Menassa, R. & Guedes, M.I.F. (2020) Transient expression of dengue virus NS1 antigen in Nicotiana benthamiana for use as a diagnostic antigen. Frontiers in Plant Science, 10, 1674.
 Go to original source...
Go to original source... - Meins, F. (2000) RNA degradation and models for post-transcriptional gene silencing. Plant Molecular Biology, 43, 261-273.
 Go to original source...
Go to original source... - Nanasato, Y., Konagaya, K., Okuzaki, A., Tsuda, M. & Tabei, Y. (2013) Improvement of Agrobacterium-mediated transformation of cucumber (Cucumis sativus L.) by combination of vacuum infiltration and co-cultivation on filter paper wicks. Plant Biotechnology Reports, 7, 267-276.
 Go to original source...
Go to original source... - Reed, J. & Osbourn, A. (2018) Engineering terpenoid production through transient expression in Nicotiana benthamiana. Plant Cell Reports, 37, 1431-1441.
 Go to original source...
Go to original source... - Rivera, A.L., Gómez-Lim, M., Fernández, F. & Loske, A.M. (2012) Physical methods for genetic plant transformation. Physics of Life Reviews, 9, 308-345.
 Go to original source...
Go to original source... - Selvaraj, N., Kasthurirengan, S., Vasudevan, A., Manickavasagam, M., Choi, C.W. & Ganapathi, A. (2010) Evaluation of green fluorescent protein as a reporter gene and phosphinothricin as the selective agent for achieving a higher recovery of transformants in cucumber (Cucumis sativus L. cv. Poinsett76) via Agrobacterium tumefaciens. In Vitro Cellular & Developmental Biology - Plant, 46, 329-337.
 Go to original source...
Go to original source... - Shibata, Y., Kawakita, K. & Takemoto, D. (2010) Age-related resistance of Nicotiana benthamiana against hemibiotrophic pathogen Phytophthora infestans requires both ethylene- and salicylic acid-mediated signaling pathways. Molecular Plant-Microbe Interactions, 23, 1130-1142.
 Go to original source...
Go to original source... - Simmons, C.W., VanderGheynst, J.S. & Upadhyaya, S.K. (2009) A model of Agrobacterium tumefaciens vacuum infiltration into harvested leaf tissue and subsequent in planta transgene transient expression. Biotechnology and Bioengineering, 102, 965-970.
 Go to original source...
Go to original source... - Spiegel, H., Schillberg, S. & Nölke, G. (2022) Production of recombinant proteins by Agrobacterium-mediated transient expression. In: Schillberg, S. & Spiegel, H. (Eds.) Recombinant Proteins in Plants. Methods in Molecular Biology. Vol. 2480. New York: Humana, pp. 89-102.
 Go to original source...
Go to original source... - Stahl, Y., Grabowski, S., Bleckmann, A. et al. (2013) Moderation of Arabidopsis root stemness by CLAVATA1 and ARABIDOPSIS CRINKLY4 receptor kinase complexes. Current Biology, 23, 362-371.
 Go to original source...
Go to original source... - Stephenson, M.J., Reed, J., Brouwer, B. & Osbourn, A. (2018) Transient expression in Nicotiana benthamiana leaves for triterpene production at a preparative scale. Journal of Visualized Experiments, 138, e58169.
 Go to original source...
Go to original source... - Tague, B.W. & Mantis, J. (2006) In planta Agrobacterium-mediated transformation by vacuum infiltration. In: Salinas, J. & Sanchez-Serrano, J.J. (Eds.) Arabidopsis Protocols. Methods in Molecular Biology. Vol. 323. Totowa: Humana Press, pp. 215-223.
 Go to original source...
Go to original source... - Trull, B.N., Sultana, M.S., Pfotenhauer, A.C. et al. (2024) Robust soybean leaf agroinfiltration. Plant Cell Reports, 43, 162.
 Go to original source...
Go to original source... - Wassenegger, M. & Pélissier, T. (1998) A model for RNA-mediated gene silencing in higher plants. Plant Molecular Biology, 37, 349-362.
 Go to original source...
Go to original source... - Wroblewski, T., Tomczak, A. & Michelmore, R. (2005) Optimization of Agrobacterium-mediated transient assays of gene expression in lettuce, tomato and Arabidopsis. Plant Biotechnology Journal, 3, 259-273.
 Go to original source...
Go to original source... - Zhang, Y., Ru, Y., Shi, Z. et al. (2023) Effects of different light conditions on transient expression and biomass in Nicotiana benthamiana leaves. Open Life Sciences, 18, 20220732.
 Go to original source...
Go to original source... - Zheng, L., Liu, G., Meng, X., Li, Y. & Wang, Y. (2012) A versatile Agrobacterium-mediated transient gene expression system for herbaceous plants and trees. Biochemical Genetics, 50, 761-769.
 Go to original source...
Go to original source...



 ORCID...
ORCID...