Biologia plantarum 70:56-64, 2026 | DOI: 10.32615/bp.2026.007
Rapid in vitro propagation of elite female plants of Idesia polycarpa var. vestita Diels
- 1 Engineering Research Center of Biomass Materials, Ministry of Education, College of Life Sciences and Agri-Forestry, Southwest University of Science and Technology, Mianyang, Sichuan 621010, P.R. China
- 2 Sichuan Academy of Forestry, Chengdu, Sichuan 610084, P.R. China
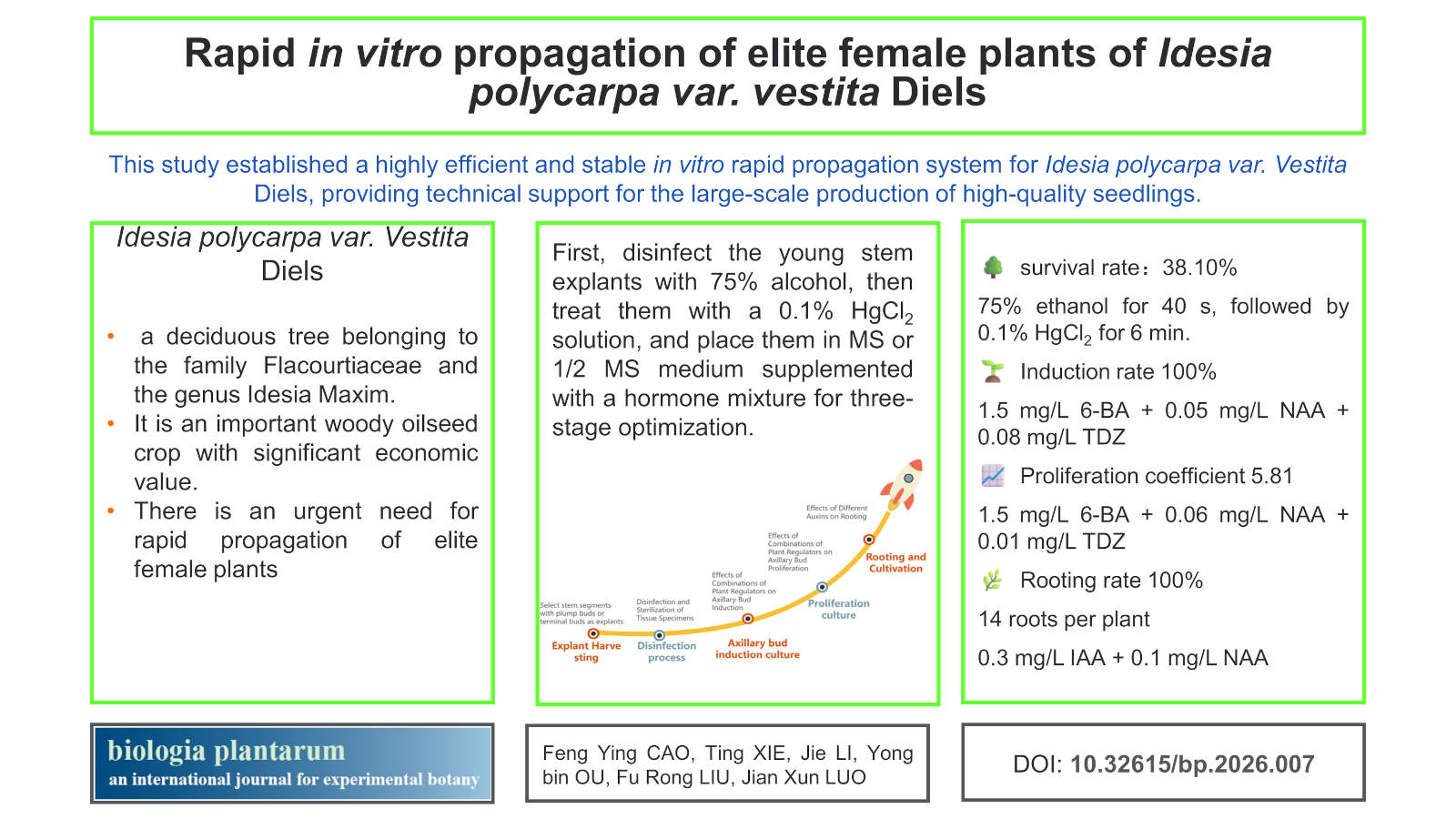
Background: Idesia polycarpa var. vestita Diels is an oil-bearing woody plant of significant economic value.
Aims: To accelerate the propagation of its elite germplasm, this study aimed to establish an efficient and stable in vitro rapid propagation system using one-year-old stem segments from elite plants.
Methods: Young stem explants were disinfected with 75% ethanol for 40 s, followed by 0.1% HgCl2 for 6 min. MS or 1/2 MS served as basal media, and various combinations of plant growth regulators were tested for axillary bud induction, proliferation, and rooting.
Results: The optimal disinfection protocol yielded a 38.10% survival rate. The best axillary bud induction medium was MS + 1.5 mg/L 6-BA + 0.05 mg/L NAA + 0.08 mg/L TDZ, achieving 100% induction. For proliferation, MS + 1.5 mg/L 6-BA + 0.06 mg/L NAA + 0.01 mg/L TDZ (proliferation coefficient 5.81) was optimal. Rooting was best with 1/2 MS + 0.3 mg/L IAA + 0.1 mg/L NAA (100% rooting, 14 roots/plant).
Conclusions: This study established a reliable micropropagation system for Idesia polycarpa var. vestita, providing technical support for the efficient production of high-quality seedlings.
Keywords: Idesia polycarpa var. vestita Diels, plant regulator, tissue culture.
Received: January 19, 2026; Revised: March 6, 2026; Accepted: March 31, 2026; Published online: April 21, 2026 Show citation
| ACS | AIP | APA | ASA | Harvard | Chicago | Chicago Notes | IEEE | ISO690 | MLA | NLM | Turabian | Vancouver |
Introduction
Idesia polycarpa var. vestita Diels (I. vestita) is a deciduous tree belonging to the genus Idesia within the Flacourtiaceae family, possessing significant economic value. Its wood is soft-textured, suitable for construction materials or furniture manufacturing. This species produces abundant, fragrant flowers with nectaries, making it a vital nectar source for beekeeping. Its elegant form is commonly cultivated as an ornamental tree in mountainous regions and gardens. The fruits and seeds of I. vestita are rich in oils, making it a high-yield, high-quality woody oil tree species and earning it the reputation as a "tree oil reservoir" (Zuo et al., 2024). Furthermore, I. vestita fruits exhibit exceptionally high levels of unsaturated fatty acids, with linoleic acid exceeding 70% (Fan et al., 2019). Purified I. vestita extracts demonstrate positive effects on cardiovascular health by reducing low-density lipoprotein cholesterol (LDL-C) levels, thereby lowering atherosclerosis risk (Della Guardia and Shin, 2022). I. vestita oil is also rich in vitamin E and polyphenolic compounds, which exhibit significant antioxidant activity, scavenging free radicals and delaying cellular aging (Liu et al., 2025). I. vestita can be utilized in industrial production of biodegradable lubricants (Li et al., 2019). I. vestita features high yield, high oil content, and excellent quality, making it an excellent choice for alleviating edible oil supply shortages and energy constraints (Wen et al., 2022). As the primary cultivated species of the genus Idesia in China, I. vestita seeds yield oil rich in linoleic acid and bioactive compounds, including β-sitosterol and tocopherols (Li et al., 2019; Song et al., 2022). Mountain jujube oil is easily digestible and absorbable, with a nutritional profile comparable to that of soybean and olive oils. It is rich in multiple components beneficial to human health and has the potential to prevent and treat various diseases (Li et al., 2020). Therefore, this tree species is regarded as a highly promising woody oilseed crop. Its potential to develop and utilize high-quality edible oils has garnered significant attention from researchers and consumers alike (Xiang et al., 2024).
If I. vestita can be mass-produced, its rapid propagation and cultivation could be widely promoted, thereby significantly advancing the development of biodiesel (Yang et al., 2009). The industrialized seedling propagation method for I. vestita in China mainly relies on seed reproduction. Currently, most practical cultivation methods utilize seedlings derived from sexual reproduction. Unfortunately, this method cannot guarantee the retention of superior parental traits, and it typically takes 4 - 5 years to differentiate male from female plants. This delay complicates the optimal arrangement of the male-to-female planting ratio, which directly influences the economic viability of subsequent stages. Therefore, the reproductive characteristics of dioecious I. vestita determine that the selection of superior varieties must be based on clone propagation (Wu et al., 2023).
Cutting, grafting, and tissue culture are the core techniques for asexual propagation of I. vestita. Previous studies have conducted experiments on hardwood and softwood cuttings of I. vestita, but the low rooting rate makes it difficult to apply and popularize on a large scale. In tissue culture studies of I. vestita, explant selection, sterilization methods, and medium formulation optimization are key factors affecting culture efficiency and regeneration quality. Hong Conghui established a simple, efficient, rapid-propagation system using young stem segments with buds from superior female plants of I. vestita as explants, providing technical support for the practical application of stem segment culture (Hong et al., 2023). Explant type directly affects callus induction efficiency, among which leaves and young roots of aseptic seedlings are widely used explant materials in I. vestita tissue culture; Fan Ruishen used leaves as explants and identified MS + 2,4-D 2.0 mg/L + 6-BA 1.0 mg/L as the optimal medium for callus induction (Fan, 2021); Shen Yangcheng used leaves as experimental materials and determined MS + NAA 1.0 mg/L as the optimal medium for callus induction (Shen, 2014); Liu Xiaomin systematically investigated the effects of explant type, plant hormone combinations, and other factors on various stages of I. vestita tissue culture using seeds and leaves as explants (Liu, 2009). Additionally, Li et al. (2014) conducted studies using young roots as explants and identified MS + TDZ 0.02 mg/L + NAA 0.5 mg/L + 6-BA 0.1 mg/L as the optimal medium formulation for callus induction.
In summary, although researchers have conducted extensive explorations on tissue culture techniques of I. vestita, systematic studies using stem segments as explants remain insufficient, and the relevant technical system has not yet been fully established. Therefore, this study employed young stem segments from 1-year-old I. vestita as explants to optimize rapid in vitro propagation technology. By screening the optimal sterilization protocol for explants and systematically investigating the effects of different plant growth regulator combinations on axillary bud induction, proliferation, and rooting, we ultimately established an efficient and stable in vitro rapid propagation system for young stem segments of I. vestita, providing theoretical support and technical guarantee for industrialized seedling production of this species.
Materials and methods
Plant material: In mid-April, the stem segments with axillary buds from female plants with high oil content, selected from the germplasm nursery of Southwest University of Science and Technology (SWUST), were collected and stored in sealed bags.
Disinfection and sterilization of explants: The annual stem segments with axillary buds were collected and placed in a beaker, rinsed with tap water for 2 h, and then transferred to a laminar flow hood. The surfaces of the explants were cleaned with 75% alcohol and 0.1% HgCl2. As shown in Table 1, the subsequent treatment durations were determined. After disinfection with 75% alcohol, the explants were rinsed three times with sterile water, surface moisture was blotted with sterile filter paper, and then they were shaken in 0.1% HgCl2 solution. Subsequently, they were rinsed with sterile water five to six times. Finally, the surface moisture of the materials was blotted with sterile filter paper. After severing both ends exposed during the disinfection procedure with the sterilized inoculation knife, the section with the axillary buds was saved, and the explants were inoculated into the MS medium. 10 - 14 stem segments were inoculated per treatment, with 3 replicates per treatment. The rates of contamination, survival, and browning were collected and computed after 10 days.
| No. | 75% alcohol (s) | 0.1% HgCl2 (min) |
|---|---|---|
| 1 | 20 | 2 |
| 2 | 20 | 4 |
| 3 | 20 | 6 |
| 4 | 40 | 2 |
| 5 | 40 | 4 |
| 6 | 40 | 6 |
| 7 | 60 | 2 |
| 8 | 60 | 4 |
| 9 | 60 | 6 |
Table 1. Different disinfection protocols for stem segments of I. vestita.
Effects of combinations of plant regulators on axillary bud induction: Three plant regulators - 6-benzylaminopurine (6-BA), α-naphthaleneacetic acid (NAA), and thidiazuron (TDZ) - were selected for an orthogonal experiment based on earlier studies that used MS as the basal medium. The concentrations of each regulator were 6-BA (mg/L): 1.0, 1.5, 2.0; NAA (mg/L): 0.03, 0.05, 0.08; and TDZ (mg/L): 0.03, 0.05, 0.08. To select the best combination of plant hormone doses for I. vestita axillary bud induction media, sterilized young stem segments with axillary buds were used for induction culture. Ten stem segments were inoculated per treatment, with three replicates for each treatment. After 30 days, the growth stage of the axillary buds was inspected, and the induction rate of the buds was tallied and determined.
Effects of combinations of plant regulators on axillary bud proliferation: For orthogonal tests, three plant regulators (6-BA, NAA, and TDZ) were chosen based on previous studies using MS as the basal medium. The concentrations of each regulator were 6-BA (mg/L): 1.0, 1.5, 2.0; NAA (mg/L): 0.03, 0.06, 0.09; TDZ (mg/L): 0.01, 0.03, 0.05. In order to choose the optimal ratio of plant hormone concentrations for the axillary bud proliferation medium of I. vestita, the robust and sterile axillary buds that had grown to a height of approximately 2 cm were excised. Five explants were inoculated per treatment, with three replicates for each treatment. The proliferation coefficient was determined after 30 days.
Following the culture of proliferation, the proliferating buds were divided into single-bud seedlings and cultured again in MS medium.
Effects of combinations of plant regulators on rooting: Based on previous studies using 1/2 MS medium as the basal medium, two plant regulators (IAA and NAA) were selected for investigation. Each hormone had the following concentrations: NAA (mg/L): 0.1, 0.3, and 0.5; IAA (mg/L): 0, 0.3, 0.5, and 0.7. Following rejuvenation culture, 2 - 3 cm sterile seedlings were used as starting material for rooting culture to determine the optimal ratio of plant hormone concentrations for the rooting medium of I. vestita. Five to six aseptic seedlings were inoculated per treatment, with three replicates for each treatment. After 30 days, the average number of roots, average root length, and rooting rate were measured and calculated.
Culture conditions: The cultivation conditions were maintained at 24 ± 2°C, with a photosynthetic photon flux density (PPFD) of 28 - 37 μmol(photon) m-2 s-1, and a photoperiod of 14 h per day.
Calculation of indices: The formulae for calculating the indices were as follows: contamination rate (%) = (number of contaminated explants/total number of explants) × 100; survival rate (%) = (number of surviving explants/total number of explants) × 100; browning rate (%) = (number of browned explants/total number of explants) × 100; induction rate (%) = (number of axillary buds induced/total number of stem segments) × 100; proliferation coefficient = (number of proliferated shoots/total number of stem segments); rooting rate (%) = (number of rooted plantlets/number of plantlets inoculated) × 100.
Data analysis: SPSS 26.0 statistical software (IBM Corp., Armonk, NY, USA) was used to process the data. Based on the results from the single-factor completely randomized design test, each index was examined using analysis of variance (ANOVA) and the F-test. Statistical correlation and significant differences were also examined. The charts were generated using the Origin software (OriginLab Corp., Northampton, MA, USA).
Results
Sterilization treatment: Fig. 1 illustrates how various combinations of disinfection durations significantly impacted the aseptic culture of stem segments. The durations of disinfection with 75% alcohol and 0.1% HgCl2 were closely related to the contamination rate, browning rate, and mortality rate of explants. Specifically, the longer the disinfection time with 75% alcohol and 0.1% HgCl2, the lower the contamination rate and the higher the browning rate, and the survival rate first increased and then decreased. In summary, the optimal disinfection method for stem segments was to disinfect with 75% alcohol for 40 s, and then disinfect with 0.1% HgCl2 for 6 min (No. 6). The contamination rate of this group was 42.86%, the survival rate was 38.10%, and the browning rate was 47.62%.
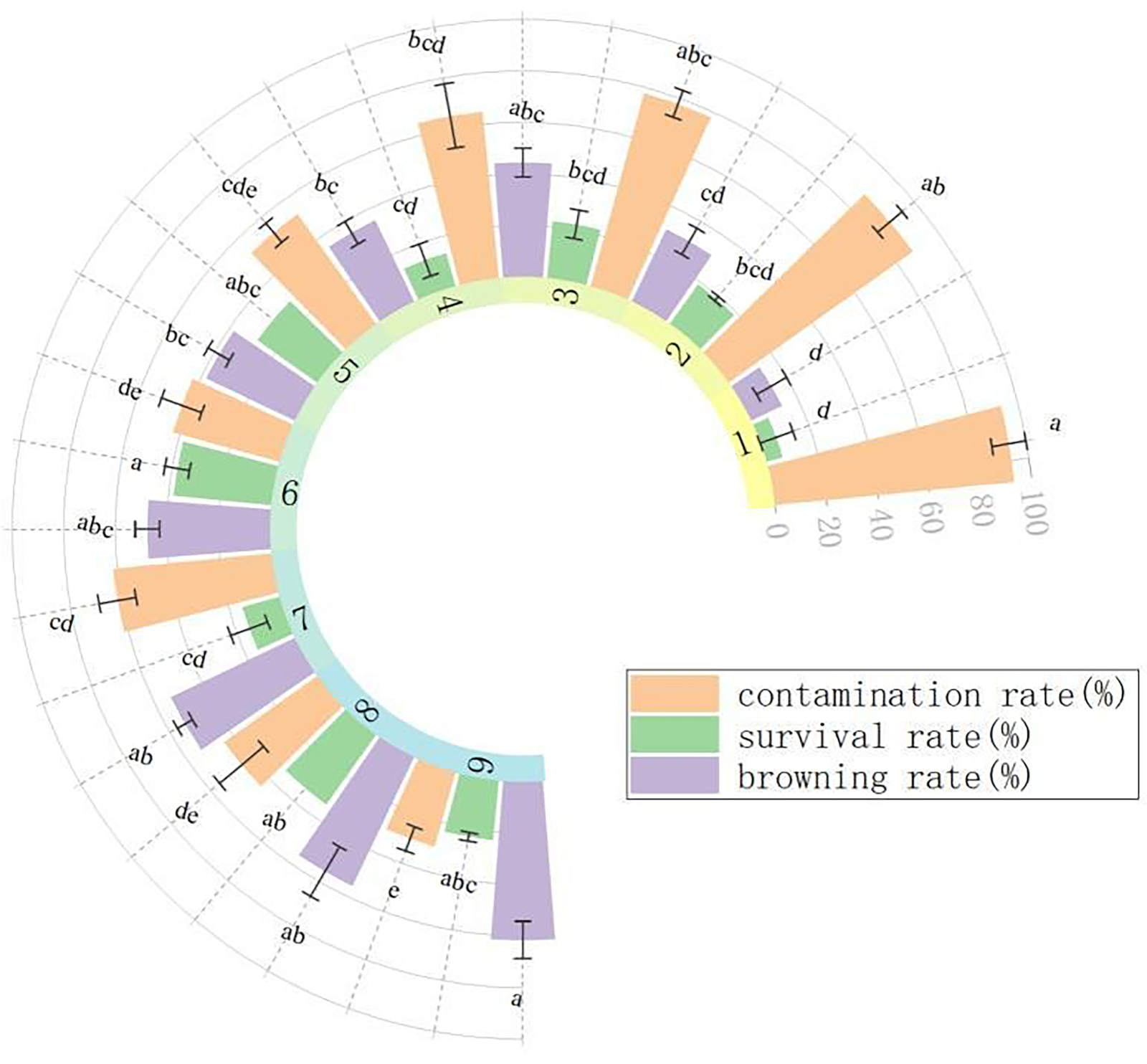
Fig. 1. Effects of different disinfection methods on the stem segments of I. vestita. Treatments 1 - 3 were disinfected with 75% ethanol for 20 s, treatments 4 - 6 for 40 s, and treatments 7 - 9 for 60 s; subsequently, treatments 1, 4, and 7 were treated with 0.1% HgCl2 for 2 min, treatments 2, 5, and 8 for 4 min, and treatments 3, 6, and 9 for 6 min. Mean ± SD, n = 3. Different letters indicate significant differences according to one-way ANOVA (P < 0.05).
Effects of plant regulators on axillary bud induction: Within 1 - 2 weeks after being transferred to the induction medium, the axillary buds began to sprout. As shown in Table 2, the effects of different concentrations of 6-BA and TDZ on axillary bud induction were significantly different (P < 0.05). In contrast, the effect of NAA on axillary bud induction was not significantly different (P > 0.05). After approximately 15 days of culture, the sprouted axillary buds could be observed; by day 15, all the axillary buds in No. 5 (1.5 mg/L 6-BA + 0.05 mg/L NAA + 0.08 mg/L TDZ) had sprouted, whereas the other groups showed only partial sprouting. By day 30, no new buds sprouted in any group. At this point, the axillary bud sprouting rates for each group were statistically assessed; No. 5 showed the highest sprouting rate (100.00%), which was significantly higher than those of the other groups. No. 4 had the lowest sprouting rate at 40.00%. Secondly, No. 2 (1.0 mg/L 6-BA + 0.05 mg/L NAA + 0.05 mg/L TDZ) had a sprouting rate of 50.00%. Additionally, the axillary buds of No. 4 (1.5 mg/L 6-BA + 0.03 mg/L NAA + 0.05 mg/L TDZ) showed poor development, with some seedlings exhibiting weak quality and yellowing leaves (Fig. 2A). In contrast, the axillary buds of No. 5 were in good growth condition, resulting in robust seedlings with deep green leaves (Fig. 2B). No. 6 (1.5 mg/L 6-BA + 0.08 mg/L NAA + 0.03 mg/L TDZ) and No. 9 (2.0 mg/L 6-BA + 0.08 mg/L NAA + 0.05 mg/L TDZ) exhibited medium induction rates with relatively strong buds (Fig. 2C, F). No. 7 (2.0 mg/L 6-BA + 0.03 mg/L NAA + 0.08 mg/L TDZ) showed an induction rate of 73.33% at 15 d and 83.33% at 30 d, but the buds were weak with light green leaves (Fig. 2D). Treatment group No. 3 - when high concentrations of TDZ are combined with high concentrations of NAA, they readily induce vitrified, malformed, or fragile adventitious buds. Due to physiological abnormalities in the later stages of culture, these buds struggle to develop into robust buds, resulting in an effective induction rate on day 30 that falls below the initial induction level observed on day 15. Additionally, in treatment group No. 8, at day 15, the hormone combination was relatively balanced, with explants at peak primordia formation, resulting in a high induced rate of statistics. By day 30, likely due to TDZ instability, the hormone balance was disrupted, preventing sustained high induction rates. Furthermore, some early-induced buds underwent callus formation and browning, resulting in a decline in the induction rate under this combination (Fig. 2E). Comprehensive analysis suggested that the optimal medium for inducing axillary buds of I. vestita was MS + 1.5 mg/L 6-BA + 0.05 mg/L NAA + 0.08 mg/L TDZ.
| Plant regulator (mg/L) | ||||||
|---|---|---|---|---|---|---|
| No. | 6-BA | NAA | TDZ | Induction rate at 15 d (%) | Induction rate at 30 d (%) | The bud status |
| 1 | 1.0 | 0.03 | 0.03 | 46.67 ± 6.67c | 66.67 ± 3.33c | Relatively strong buds and light green leaves |
| 2 | 1.0 | 0.05 | 0.05 | 30.00 ± 5.77c | 50.00 ± 5.77d | Relatively weak buds and light green leaves |
| 3 | 1.0 | 0.08 | 0.08 | 73.33 ± 8.82b | 66.67 ± 3.33c | Relatively strong buds and light green leaves |
| 4 | 1.5 | 0.03 | 0.05 | 33.33 ± 3.33c | 40.00 ± 5.77d | Weak buds and yellow leaves |
| 5 | 1.5 | 0.05 | 0.08 | 100.00 ± 0.00a | 100.00 ± 0.00a | Strong buds and deep green leaves |
| 6 | 1.5 | 0.08 | 0.03 | 73.33 ± 3.33b | 76.67 ± 3.33bc | Relatively strong buds and light green leaves |
| 7 | 2.0 | 0.03 | 0.08 | 73.33 ± 6.67b | 83.33 ± 3.33b | Weak buds and light green leaves |
| 8 | 2.0 | 0.05 | 0.03 | 80.00 ± 5.77b | 76.67 ± 3.33bc | Strong buds and deep green leaves |
| 9 | 2.0 | 0.08 | 0.05 | 76.67 ± 8.82b | 80.00 ± 5.77bc | Relatively strong buds and deep green leaves |
Table 2. Effects of combinations of plant regulators on axillary bud induction. Mean ± SD, n = 3. Different letters indicate significant differences according to one-way ANOVA (P < 0.05).

Fig. 2. Axillary bud induction at 30 d. A: No. 4 (1.5 mg/L 6-BA + 0.03 mg/L NAA + 0.05 mg/L TDZ); B: No. 5 (1.5 mg/L 6-BA + 0.05 mg/L NAA + 0.08 mg/L TDZ); C: No. 6 (1.5 mg/L 6-BA + 0.08 mg/L NAA + 0.03 mg/L TDZ); D: No. 7 (2.0 mg/L 6-BA + 0.03 mg/L NAA + 0.08 mg/L TDZ); E: No. 8 (2.0 mg/L 6-BA + 0.05 mg/L NAA + 0.03 mg/L TDZ); F: No. 9 (2.0 mg/L 6-BA + 0.08 mg/L NAA + 0.05 mg/L TDZ).
Effects of plant regulators on axillary bud proliferation: After being transferred to the propagation medium, the buds sprouted lateral shoots under the regulator's influence, as shown in Table 3, which summarizes the propagation coefficients after 30 days. There were significant differences in axillary bud growth in response to the concentrations of 6-BA, NAA, and TDZ. The No. 5 (1.5 mg/L 6-BA + 0.06 mg/L NAA + 0.01 mg/L TDZ) axillary buds exhibited the highest proliferation coefficient at 5.81, with vigorous growth and deep green leaves (Fig. 3A). No. 3 (1.0 mg/L 6-BA + 0.09 mg/L NAA + 0.05 mg/L TDZ) showed a medium proliferation coefficient of 2.71, with general growth status and green leaves (Fig. 3B). No. 6 (1.5 mg/L 6-BA + 0.09 mg/L NAA + 0.05 mg/L TDZ) had the lowest coefficient at 2.1, showing poor development of the growth and yellowish leaves, with some seedlings exhibiting vitrification (Fig. 3C). Additionally, there was a statistically significant difference (P < 0.05) in the proliferation coefficient of germination for No. 5 compared to other groups. Comprehensive analysis indicated that the optimal medium for axillary bud growth of I. vestita was MS + 1.5 mg/L 6-BA + 0.06 mg/L NAA + 0.01 mg/L TDZ.
| Plant regulator (mg/L) | |||||
|---|---|---|---|---|---|
| No. | 6-BA | NAA | TDZ | Proliferation coefficient | Growth status |
| 1 | 1.0 | 0.03 | 0.01 | 2.86 ± 0.19cd | General growth with green leaves |
| 2 | 1.0 | 0.06 | 0.03 | 2.99 ± 0.52cd | Fine growth with green leaves |
| 3 | 1.0 | 0.09 | 0.05 | 2.71 ± 0.32cd | General growth with green leaves |
| 4 | 1.5 | 0.03 | 0.03 | 4.27 ± 0.15b | General growth with green leaves |
| 5 | 1.5 | 0.06 | 0.01 | 5.81 ± 0.67a | Fine growth with green leaves |
| 6 | 1.5 | 0.09 | 0.05 | 2.10 ± 0.10d | Bad growth with yellow leaves |
| 7 | 2.0 | 0.03 | 0.05 | 3.22 ± 0.22c | Fine growth with green leaves |
| 8 | 2.0 | 0.06 | 0.01 | 2.83 ± 0.17cd | Fine growth with green leaves |
| 9 | 2.0 | 0.09 | 0.03 | 2.89 ± 0.11cd | General growth with green leaves |
Table 3. Effects of combinations of plant regulators on axillary bud proliferation at 30 d. Mean ± SD, n = 3. Different letters indicate significant differences according to one-way ANOVA (P < 0.05).

Fig. 3. Observation of axillary bud proliferation at 30 d. A: No. 5 (1.5 mg/L 6-BA + 0.06 mg/L NAA + 0.01 mg/L TDZ); B: No. 3 (1.0 mg/L 6-BA + 0.09 mg/L NAA + 0.05 mg/L TDZ); C: No. 6 (1.5 mg/L 6-BA + 0.09 mg/L NAA + 0.05 mg/L TDZ).
Effects of different auxins on rooting: Seedlings of I. vestita were transplanted into the rooting medium. New root formation was observed during the 2 - 3 weeks, while lateral roots were observed after 4 weeks. Table 4 presents the results of seedling rooting. The combinations of IAA and NAA significantly affected the rooting rate of I. vestita (P < 0.05). Group 4 (0.3 mg/L IAA + 0.1 mg/L NAA) achieved the highest rooting rate (100%), significantly higher than other treatment groups (P < 0.05). Group 5 (0.3 mg/L IAA + 0.3 mg/L NAA) ranked second. Group 12 (0.7 mg/L IAA + 0.5 mg/L NAA) exhibited the lowest rooting rate (26.67%), which was significantly lower than that of the CK group (P < 0.05). The combinations of IAA and NAA also had a significant impact on the average number of roots of I. vestita (P < 0.05). The average number of roots per plant was the highest in No. 4, with 14.00 roots.
| Plant regulator (mg/L) | Rooting status | ||||
|---|---|---|---|---|---|
| No. | IAA | NAA | Rooting rate (%) | Average length of roots per plant (cm) | Average number of roots per plant |
| CK | 0 | 0 | 43.33 ± 3.33ef | 2.72 ± 0.15cd | 6.33 ± 0.67cd |
| 1 | 0 | 0.1 | 56.67 ± 3.33d | 1.88 ± 0.32fg | 5.00 ± 0.41cd |
| 2 | 0 | 0.3 | 53.33 ± 3.33de | 2.62 ± 0.14de | 5.40 ± 0.51cd |
| 3 | 0 | 0.5 | 33.33 ± 3.33fg | 1.98 ± 0.25efg | 5.00 ± 0.00cd |
| 4 | 0.3 | 0.1 | 100.00 ± 0.00a | 4.84 ± 0.38a | 14.00 ± 1.38a |
| 5 | 0.3 | 0.3 | 96.67 ± 3.33a | 3.60 ± 0.20b | 11.25 ± 0.85b |
| 6 | 0.3 | 0.5 | 56.67 ± 3.33d | 2.88 ± 0.10cd | 7.00 ± 1.00c |
| 7 | 0.5 | 0.1 | 83.33 ± 3.33b | 3.72 ± 0.31b | 7.00 ± 1.10c |
| 8 | 0.5 | 0.3 | 73.33 ± 3.33c | 3.36 ± 0.19bc | 10.60 ± 0.51b |
| 9 | 0.5 | 0.5 | 40.00 ± 5.77f | 1.58 ± 0.20gh | 4.50 ± 0.50cd |
| 10 | 0.7 | 0.1 | 53.33 ± 3.33de | 2.46 ± 0.09def | 3.67 ± 0.33de |
| 11 | 0.7 | 0.3 | 40.00 ± 0.00f | 2.48 ± 0.15def | 5.33 ± 0.33cd |
| 12 | 0.7 | 0.5 | 26.67 ± 3.33g | 1.12 ± 0.13h | 1.50 ± 0.50e |
Table 4. Effects of different auxins on rooting at 30 d. Mean ± SD, n = 3. Different letters indicate significant differences according to one-way ANOVA (P < 0.05). CK - hormone-free medium.
The average number of roots for No. 12 was the lowest at 1.50, showing a significant difference compared with the blank control group (P < 0.05). The blank control group without exogenous plant growth regulators (Fig. 4A, F) had an average root number of 6.33, with poor root development and slender morphology. No. 2 supplemented with 0.3 mg/L NAA alone had an average root number of 5.40, which was lower than that of the control group, indicating that NAA alone failed to promote root number increase, and root growth remained unsatisfactory (Fig. 4D, I). The concentrations of IAA and NAA significantly affected the average root length of I. vestita (P < 0.05). No. 4 had the highest average root length at 4.84 cm. No. 12 had the shortest average root length at 1.12 cm, followed by No. 9 (0.5 mg/L IAA + 0.5 mg/L NAA) at 1.58 cm. As shown in Table 4, IAA was the primary hormone influencing rooting rate, average root number, and average root length. As IAA hormone concentration increased, the rooting rate, average number of roots, and average root length showed a pattern of first rising and then falling. At a concentration of 0.3 mg/L (Fig. 4B, G), the root length was the largest, and the main root was slender; at 0.5 mg/L IAA (Fig. 4C, H), the root length was moderate; at a concentration of 0.7 mg/L (Fig. 4E, J), the root length was minimized, and there were fewer lateral roots. Comprehensive consideration, 1/2 MS + 0.3 mg/L IAA + 0.1 mg/L NAA was the ideal rooting medium for I. vestita.

Fig. 4. Observation of strong and rooted seedlings at 30 d. A: CK - hormone-free medium; B: No. 4 (0.3 mg/L IAA + 0.1 mg/L NAA); C: No. 7 (0.5 mg/L IAA + 0.1 mg/L NAA); D: No. 2 (0.3 mg/L NAA); E: No. 12 (0.7 mg/L IAA + 0.5 mg/L NAA); Sections F - J correspond to A - E.
Discussion
Currently, there is a limited research on the rapid propagation technology of I. vestita stem segments, making it difficult to meet the need for rapid, efficient reproduction. In this study, I. vestita stem segments with buds were used as explants to construct an in vitro rapid propagation system, aiming to provide technical support for the industrial production of high-quality I. vestita seedlings.
Effects of explant disinfection on in vitro rapid propagation system: The results of this experimental study indicate that the optimal sterilization treatment for one-year-old stem segments of I. vestita involves first treating them with 75% ethanol for 40 s, followed by treatment with 0.1% HgCl2 for 6 min. This protocol achieved contamination rates of 42.86%, survival rates of 38.10%, and browning rates of 47.62%. However, overall contamination and browning rates remained high, and survival rates were low.
Establishing a sterile tissue culture system is fundamental to developing a rapid plant propagation system. The core challenge lies in thoroughly eliminating microbial contamination from explant surfaces while preserving explant physiological activity to the greatest extent possible (Han et al., 2023). I. vestita stem segments feature densely distributed glandular trichomes on their surfaces, presenting greater challenges for establishing sterility compared to smoother explants. This study demonstrates that as disinfectant exposure time increases, contamination rates decrease, survival rates initially rise, then decline, and browning rates increase. This indicates that when disinfectant sterilization time is excessively prolonged, alcohol and HgCl2 not only eliminate microorganisms but also penetrate plant tissues, where their toxicity inhibits normal growth (Gammoudi et al., 2022). Wang Haiyang similarly observed in his explant sterilization research that prolonged immersion of explants in 0.1% HgCl2 caused greater damage to explants (Wang, 2015). Barton and Koricheva (2010) found that woody plants contain significantly higher levels of phenolic compounds than herbaceous plants. Substances with high phenolic and quinone content typically exhibit more pronounced browning issues during tissue culture. Browning refers to the process where explants produce and release ketone-containing substances during tissue culture. These substances diffuse into the medium, gradually turning it brown and inhibiting the activity of other enzymes, leading to browning and death of the explants (Permadi et al., 2024). Therefore, adding antioxidants such as ascorbic acid and activated charcoal to the culture medium can reduce browning rates in woody plants during tissue culture.
Effects of plant regulator on in vitro rapid propagation system: The results of this study demonstrate that plant growth regulators play a crucial role in axillary bud induction, proliferation, and rooting in the micropropagation system of I. vestita. Specifically, the optimal combination for axillary bud induction was 1.5 mg/L 6-BA, 0.05 mg/L NAA, and 0.08 mg/L TDZ, resulting in a 100% induction rate. For axillary bud proliferation, the optimal combination was 1.5 mg/L 6-BA, 0.06 mg/L NAA, and 0.01 mg/L TDZ, yielding a proliferation coefficient of 5.81 and robust bud growth. The most effective combination for rooting was 0.3 mg/L IAA and 0.1 mg/L NAA, resulting in a 100% rooting rate, with an average of 14.0 roots per plantlet and an average root length of 4.8 cm.
Plant growth regulators come in a wide variety, among which TDZ exhibits exceptionally potent cytokinin activity, effectively inducing adventitious bud differentiation. Among these, TDZ initiates morphogenesis through metabolic cascades and stress responses, establishing a dual-action mechanism (Jones et al., 2007). However, its sole application often results in vitrification and deformities in tissue-cultured seedlings (Huetteman and Preece, 1993). In contrast, 6-BA acts mildly and stably, promoting bud proliferation and robust seedling development. The combined use of these two compounds achieves complementary advantages. Nie Wangxing's research indicates that the combined application of 0.5 mg/L 6-BA and 1.0 mg/L TDZ results in the highest tillering induction rate and the greatest number of shoots per cotyledon node, reaching 84.75% and 3.04, respectively (Nie and Yu, 2012). Additionally, Ren Pengbin's research demonstrates that the combined use of 6-BA and TDZ induces adventitious bud formation in konjac callus tissue (Ren et al., 2014). Therefore, in the sterile micropropagation system established in this study, various concentrations of TDZ, 6-BA, and NAA were tested in combination to identify the optimal mixture of plant growth regulators for axillary bud induction in I. vestita. Experimental results indicate that supplementation of MS medium with 1.5 mg/L 6-BA, 0.05 mg/L NAA, and 0.08 mg/L TDZ constitutes the optimal formulation for inducing axillary bud growth in I. vestita. Among these regulators, the effect on axillary bud induction follows the order: 6-BA > NAA > TDZ. Induction rates were relatively low at both lower (1.0 mg/L) and higher (2.0 mg/L) concentrations of 6-BA. The combination of 1.5 mg/L 6-BA and lower concentrations of NAA produced a synergistic effect, promoting efficient differentiation of axillary primordia and the induction of adventitious buds. This hormonal regulation pattern is consistent with findings from studies on the induction of clustered buds in Shennong fragrant chrysanthemum (Ma et al., 2016) and poplar (Zhou et al., 2012), supporting the existence of a common mechanism underlying axillary bud activation in plants.
Proliferation culture is a critical step in in vitro rapid propagation. Research indicates that the optimal combination of plant growth regulators for I. vestita proliferation culture is 1.5 mg/L 6-BA, 0.06 mg/L NAA, and 0.01 mg/L TDZ. In the experiment, the influence of 6-BA, NAA, and TDZ on axillary bud induction was observed in the following order: 6-BA > TDZ > NAA. Among these, both low (1.0 mg/L) and high (2.0 mg/L) concentrations of 6-BA resulted in relatively lower proliferation coefficient. 6-BA and TDZ significantly affect the proliferation of I. vestita, an effect also reported in studies investigating the effects of plant growth regulators on the proliferation of the red European pear cultivar 'Red Star' (Sun et al., 2020).
Rooting culture is a crucial stage in the micropropagation process. Experimental results indicate that the optimal combination of plant growth regulators for rooting is 0.3 mg/L IAA and 0.1 mg/L NAA. NAA and IAA are widely used in in vitro rapid propagation systems and rooting induction experiments for cuttings. Experimental results demonstrate that the synergistic effect of low concentrations of IAA and NAA is significantly superior to that of either regulator alone in promoting root development. This finding is consistent with results from rooting studies on Chinese fir (Wen et al., 2016). In rooting experiments, when IAA concentration exceeds a threshold, it not only reduces rooting efficiency but also reduces the number of lateral roots in I. vestita. This phenomenon also occurs during the rooting of Phalaenopsis maculata seedlings (Yuan et al., 2024).
Conclusions
This study established a sterile system for I. vestita by immersing stem segments in 75% ethanol for 40 s, followed by oscillatory immersion in 0.1% HgCl2 for 6 min. The contamination rate was 42.86%, the survival rate was 38.10%, and the browning rate was 47.62%. By determining the optimal concentrations and ratios of plant regulators for axillary bud induction, bud proliferation, and seedling rooting, an efficient in vitro rapid-propagation system for I. vestita was successfully established. The bud induction rate reached 100%, with a proliferation coefficient of 5.81. The rooting rate also reached 100%, with an average of 14.00 roots per plant and an average root length of 4.84 cm.
Download citation
References
- Barton, K.E. & Koricheva, J. (2010) The ontogeny of plant defense and herbivory: characterizing general patterns using meta-analysis. The American Naturalist, 175, 483-497.
 Go to original source...
Go to original source... - Della Guardia, L. & Shin, A.C. (2022) The role of adipose tissue dysfunction in PM2.5-induced vascular pathology. American Journal of Physiology: Heart and Circulatory Physiology, 322, H971-H972.
 Go to original source...
Go to original source... - Fan, R.S. (2021) [Cloning and functional analysis of genes related to oil synthesis in Idesia polycarpa.] PhD Thesis. Yangling: Northwest A&F University, pp. 152. [In Chinese]
- Fan, R.S., Li, L., Cai, G. et al. (2019) Molecular cloning and function analysis of FAD2 gene in Idesia polycarpa. Phytochemistry, 168, 112114.
 Go to original source...
Go to original source... - Gammoudi, N., Nagaz, K. & Ferchichi A. (2022) Establishment of optimized in vitro disinfection protocol of Pistacia vera L. explants mediated a computational approach: multilayer perceptron-multi-objective genetic algorithm. BMC Plant Biology, 22, 324.
 Go to original source...
Go to original source... - Han, S., Liu, M., Wang, Y. & Chen, J. (2023) Tissue culture and rapid propagation technology for Gentiana rhodantha. Open Life Sciences, 18, 20220565.
 Go to original source...
Go to original source... - Hong, C.H., Yu, Q., Leng, Y. et al. (2023) [Study on tissue culture and rapid propagation technology of excellent female Idesia polycarpa.] Journal of Hubei Minzu University (Natural Science Edition), 41, 141-146+155. [In Chinese]
- Huetteman, C.A. & Preece, J.E. (1993) Thidiazuron: a potent cytokinin for woody plant tissue culture. Plant Cell, Tissue and Organ Culture, 33, 105-119.
 Go to original source...
Go to original source... - Jones, M.P.A., Cao, J., O'Brien, R., Murch, S.J. & Saxena, P.K. (2007) The mode of action of thidiazuron: auxins, indoleamines, and ion channels in the regeneration of Echinacea purpurea L. Plant Cell Reports, 26, 1481-1490.
 Go to original source...
Go to original source... - Li, N., Sun, Y.-R., He, L.-B. et al. (2020) Amelioration by Idesia polycarpa Maxim. var. vestita Diels. of oleic acid-induced nonalcoholic fatty liver in HepG2 cells through antioxidant and modulation of lipid metabolism. Oxidative Medicine and Cellular Longevity, 2020, 1208726.
 Go to original source...
Go to original source... - Li, X.Y., Wang, Y.J., Wang, H.D., Wang, B. & Qu, W.Q. (2014) [Study on in vitro culture technology of young roots of Idesia polycarpa.] Anhui Agricultural Science Bulletin, 20, 22-23. [In Chinese]
- Li, Y., Peng, T., Huang, L., Zhang, S., He, Y. & Tang, L. (2019) The evaluation of lipids raw material resources with the fatty acid profile and morphological characteristics of Idesia polycarpa Maxim. var. vestita Diels fruit in harvesting. Industrial Crops and Products, 129, 114-122.
 Go to original source...
Go to original source... - Liu, F., Xie, Y., Geng, X. et al. (2025) The development and application of compound formulation of natural antioxidants in Idesia polycarpa Maxim. oil. Heliyon, 11, e41648.
 Go to original source...
Go to original source... - Liu, X.M. (2009) [Study on propagation and tissue culture technology of Idesia polycarpa.] MSc Thesis. Yangling: Northwest A&F University, pp. 51. [In Chinese]
- Ma, J., Wang, C.L., Xie, P.P., Wang, J. & Liang, H.W. (2016) [Induction of cluster buds and plant regeneration of Dendranthema indicum var. aromaticum.] Chinese Journal of Tropical Crops, 37, 505-509. [In Chinese]
- Nie, W.X. & Yu, B.J. (2012) [Effects of TDZ and 6-BA on cluster bud induction in soybean cotyledonary node regeneration system.] Journal of Nanjing Agricultural University, 35, 130-134. [In Chinese]
- Permadi, N., Akbari, S.I., Prismantoro, D. et al. (2024) Traditional and next-generation methods for browning control in plant tissue culture: Current insights and future directions. Current Plant Biology, 38, 100339.
 Go to original source...
Go to original source... - Ren, P.B., Wang, A.J., Liu, J.C. & Hu, X.P. (2014) [Effects of TDZ and 6-BA on adventitious bud induction of konjac.] Heilongjiang Agricultural Sciences, 8, 23-27. [In Chinese]
- Shen, Y.C. (2014) [Study on efficient germination and in vitro rapid propagation technology of Idesia polycarpa.] MSc Thesis. Hefei: Anhui University, pp. 60. [In Chinese]
- Song, M.F., Yang, Y., Bai, R.R. et al. (2022) [Quality and antioxidant activity in vitro of Idesia polycarpa Maxim. oils extracted by different methods.] China Condiment, 47, 28-32+38. [In Chinese]
- Sun, Q.R., Guan, Q.Z., Tao, J.H. & Sun, H.Y. (2020) [Tissue culture and adventitious shoot regeneration from in vitro leaves of red European pear cultivar 'Hongxing'.] Plant Physiology Journal, 56, 771-778. [In Chinese]
- Wang, H.Y. (2015) [Study on asexual propagation technology of Idesia polycarpa.] MSc Thesis. Zhengzhou: Henan Agricultural University, pp. 52. [In Chinese]
- Wen, L.Y., Xiang, X.W., Wang, Z.R. et al. (2022) Evaluation of cultivars diversity and lipid composition properties of Idesia polycarpa var. vestita Diels. Journal of Food Science, 87, 3841-3855.
 Go to original source...
Go to original source... - Wen, Q.L., Qin, L.F., Li, Y.M. & Mo, X.F. (2016) [Effects of IAA and NAA on rooting of Cunninghamia lanceolata tissue culture seedlings.] Hubei Agricultural Sciences, 55, 3720-3722+3740. [In Chinese]
- Wu, L.L., Deng, W.F., Lu, X.Y., Niu, C.T., Tian, H. & Li, Z. (2023) [Progress in development and utilization of Idesia polycarpa.] Nonwood Forest Research, 41, 242-252. [In Chinese]
- Xiang, X.W., Yang, Q.Q., Chen, K.W. et al. (2024) Characterization of key bitter compounds in Idesia polycarpa var. vestita Diels fruit by sensory-guided fractionation. Food Chemistry, 439, 138103.
 Go to original source...
Go to original source... - Yang, F.-X., Su, Y.-Q., Li, X.-H., Zhang, Q. & Sun, R.-C. (2009) Preparation of biodiesel from Idesia polycarpa var. vestita fruit oil. Industrial Crops and Products, 29, 622-628.
 Go to original source...
Go to original source... - Yuan, Y.H., Lei, L.L. & Ren, Y. (2024) [Effects of different media and NAA concentrations on rooting of strong seedlings of Phalaenopsis.] Modern Horticulture, 47, 10-12+16. [In Chinese]
- Zhou, Y., Gao, Z., Gao, S., Sun, F., Cheng, P. & Li, F. (2012) In vitro adventitious shoot regeneration via indirect organogenesis from inflorescence explants and peroxidase involvement in morphogenesis of Populus euphratica Olivier. Applied Biochemistry and Biotechnology, 168, 2067-2078.
 Go to original source...
Go to original source... - Zuo, Y., Liu, H., Li, B. et al. (2024) The Idesia polycarpa genome provides insights into its evolution and oil biosynthesis. Cell Reports, 43, 113909.
 Go to original source...
Go to original source...



 ORCID...
ORCID...